Boxed Warning
WARNING: RISK OF HEPATITIS B VIRUS REACTIVATION IN PATIENTS COINFECTED WITH HCV AND HBV
Test all patients for evidence of current or prior hepatitis B virus (HBV) infection before initiating treatment with EPCLUSA. HBV reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals and were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure, and death. Monitor HCV/HBV coinfected patients for hepatitis flare or HBV reactivation during HCV treatment and post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated [see Warnings and Precautions (5.1)].
Recent Major Changes
1. Indications and Usage
EPCLUSA is indicated for the treatment of adults and pediatric patients 3 years of age and older with chronic hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5, or 6 infection [see Dosage and Administration (2.2, 2.3, 2.4) and Clinical Studies (14)]:
- without cirrhosis or with compensated cirrhosis
- with decompensated cirrhosis for use in combination with ribavirin.
2. Dosage and Administration
2.1 Testing Prior to the Initiation of Therapy
Test all patients for evidence of current or prior HBV infection by measuring hepatitis B surface antigen (HBsAg) and hepatitis B core antibody (anti-HBc) before initiating HCV treatment with EPCLUSA [see Warnings and Precautions (5.1)].
2.2 Recommended Treatment Regimen and Duration in Patients 3 Years of Age and Older
Table 1 shows the recommended treatment regimen and duration based on patient population.
For patients with HCV/HIV-1 coinfection follow the dosage recommendations in Table 1. For treatment-naïve and treatment-experienced liver transplant recipients without cirrhosis or with compensated cirrhosis (Child-Pugh A), the recommended regimen is EPCLUSA once daily for 12 weeks [see Clinical Studies (14.3 and 14.5)]. Refer to Drug Interactions (7) for dosage recommendations for concomitant drugs.
| Patient Population | Treatment Regimen and Duration |
|---|---|
| Treatment-naïve and treatment-experienced*, without cirrhosis and with compensated cirrhosis (Child-Pugh A) | EPCLUSA 12 weeks |
| Treatment-naïve and treatment-experienced*, with decompensated cirrhosis (Child-Pugh B or C) | EPCLUSA + ribavirin† 12 weeks |
| |
2.3 Recommended Dosage in Adults
The recommended dosage of EPCLUSA in adults is one tablet (400 mg sofosbuvir and 100 mg velpatasvir) taken orally once daily with or without food [see Clinical Pharmacology (12.3)].
When administered with EPCLUSA, the recommended dosage of ribavirin is based on weight (administered with food): 1,000 mg per day for patients less than 75 kg and 1,200 mg for those weighing at least 75 kg, divided and administered twice daily. The starting dosage and on-treatment dosage of ribavirin can be decreased based on hemoglobin and creatinine clearance. For ribavirin dosage modifications refer to the ribavirin prescribing information [see Use in Specific Populations (8.6) and Clinical Studies (14.4)].
2.4 Recommended Dosage in Pediatric Patients 3 Years of Age and Older
The recommended dosage of EPCLUSA in pediatric patients 3 years of age and older is based on weight and provided in Table 2. Table 3 provides the weight-based dosage of ribavirin when used in combination with EPCLUSA for pediatric patients. Take EPCLUSA oral pellets or tablets once daily with or without food. In pediatric patients less than 6 years of age, administer the oral pellets with food to increase tolerability related to palatability [see Use in Specific Populations (8.4), Clinical Pharmacology (12.3), and Clinical Studies (14.8)].
| Body Weight (kg) | EPCLUSA Daily Dose | Dosing of EPCLUSA Oral Pellets | Dosing of EPCLUSA Tablet |
|---|---|---|---|
| less than 17 | 150 mg/37.5 mg per day | one 150 mg/37.5 mg packet of pellets once daily | N/A |
| 17 to less than 30 | 200 mg/50 mg per day | one 200 mg/50 mg packet of pellets once daily | one 200 mg/50 mg tablet once daily |
| at least 30 | 400 mg/100 mg per day | two 200 mg/50 mg packets of pellets once daily | one 400 mg/100 mg tablet once daily* |
| |||
| Body Weight (kg) | Oral Ribavirin Daily Dosage* |
|---|---|
| less than 47 | 15 mg per kg per day (divided dose AM and PM) |
| 47–49 | 600 mg per day (1 × 200 mg AM, 2 × 200 mg PM) |
| 50–65 | 800 mg per day (2 × 200 mg AM, 2 × 200 mg PM) |
| 66–80 | 1,000 mg per day (2 × 200 mg AM, 3 × 200 mg PM) |
| greater than 80 | 1,200 mg per day (3 × 200 mg AM, 3 × 200 mg PM) |
| |
2.5 Preparation and Administration of Oral Pellets
See the EPCLUSA oral pellets full Instructions for Use for details on the preparation and administration of EPCLUSA oral pellets.
Do not chew EPCLUSA oral pellets to avoid a bitter aftertaste. EPCLUSA oral pellets can be taken directly in the mouth or with food (See Instructions for Use). In pediatric patients less than 6 years of age, administer the oral pellets with food to increase tolerability related to palatability. Sprinkle the oral pellets on one or more spoonfuls of non-acidic soft food at or below room temperature. Examples of non-acidic foods include pudding, chocolate syrup, and ice cream. Take EPCLUSA oral pellets within 15 minutes of gently mixing with food and swallow the entire contents without chewing.
2.6 Renal Impairment
No dosage adjustment of EPCLUSA is recommended in patients with any degree of renal impairment, including patients requiring dialysis. Administer EPCLUSA with or without ribavirin according to the recommendations in Table 1 [see Adverse Reactions (6.1), Use in Specific Populations (8.6), and Clinical Studies (14.6)]. Refer to ribavirin tablet prescribing information for ribavirin dosage modification for patients with CrCl less than or equal to 50 mL per minute.
3. Dosage Forms and Strengths
EPCLUSA is available as tablets or pellets for oral use. Each dosage form is available in two dose strengths:
- 400 mg/100 mg Tablets: pink, diamond-shaped, film-coated tablet debossed with "GSI" on one side and "7916" on the other side. Each tablet contains 400 mg of sofosbuvir and 100 mg of velpatasvir.
- 200 mg/50 mg Tablets: pink, oval-shaped, film-coated tablet debossed with "GSI" on one side and "S/V" on the other side. Each tablet contains 200 mg of sofosbuvir and 50 mg of velpatasvir.
- 200 mg/50 mg Oral Pellets: white to off-white, film-coated pellets in unit-dose packets. Each packet contains 200 mg of sofosbuvir and 50 mg of velpatasvir.
- 150 mg/37.5 mg Oral Pellets: white to off-white, film-coated pellets in unit-dose packets. Each packet contains 150 mg of sofosbuvir and 37.5 mg of velpatasvir.
4. Contraindications
EPCLUSA and ribavirin combination regimen is contraindicated in patients for whom ribavirin is contraindicated. Refer to the ribavirin prescribing information for a list of contraindications for ribavirin [see Dosage and Administration (2.2, 2.3, 2.4)].
5. Warnings and Precautions
5.1 Risk of Hepatitis B Virus Reactivation in Patients Coinfected with HCV and HBV
Hepatitis B virus (HBV) reactivation has been reported in HCV/HBV coinfected patients who were undergoing or had completed treatment with HCV direct acting antivirals, and who were not receiving HBV antiviral therapy. Some cases have resulted in fulminant hepatitis, hepatic failure, and death. Cases have been reported in patients who are HBsAg positive and also in patients with serologic evidence of resolved HBV infection (i.e., HBsAg negative and anti-HBc positive). HBV reactivation has also been reported in patients receiving certain immunosuppressants or chemotherapeutic agents; the risk of HBV reactivation associated with treatment with HCV direct-acting antivirals may be increased in these patients.
HBV reactivation is characterized as an abrupt increase in HBV replication manifesting as a rapid increase in serum HBV DNA level. In patients with resolved HBV infection, reappearance of HBsAg can occur. Reactivation of HBV replication may be accompanied by hepatitis, i.e., increases in aminotransferase levels and, in severe cases, increases in bilirubin levels, liver failure, and death can occur.
Test all patients for evidence of current or prior HBV infection by measuring HBsAg and anti-HBc before initiating HCV treatment with EPCLUSA. In patients with serologic evidence of HBV infection, monitor for clinical and laboratory signs of hepatitis flare or HBV reactivation during HCV treatment with EPCLUSA and during post-treatment follow-up. Initiate appropriate patient management for HBV infection as clinically indicated.
5.2 Serious Symptomatic Bradycardia When Coadministered with Amiodarone
Postmarketing cases of symptomatic bradycardia and cases requiring pacemaker intervention have been reported when amiodarone is coadministered with a sofosbuvir-containing regimen. A fatal cardiac arrest was reported in a patient taking amiodarone who was coadministered a sofosbuvir-containing regimen (HARVONI® [ledipasvir/sofosbuvir]). Bradycardia has generally occurred within hours to days, but cases have been observed up to 2 weeks after initiating HCV treatment. Patients also taking beta blockers, or those with underlying cardiac comorbidities and/or advanced liver disease may be at increased risk for symptomatic bradycardia with coadministration of amiodarone. Bradycardia generally resolved after discontinuation of HCV treatment. The mechanism for this effect is unknown.
Coadministration of amiodarone with EPCLUSA is not recommended. For patients taking amiodarone who have no other alternative viable treatment options and who will be coadministered EPCLUSA:
- Counsel patients about the risk of symptomatic bradycardia.
- Cardiac monitoring in an in-patient setting for the first 48 hours of coadministration is recommended, after which outpatient or self-monitoring of the heart rate should occur on a daily basis through at least the first 2 weeks of treatment.
Patients who are taking EPCLUSA who need to start amiodarone therapy due to no other alternative viable treatment options should undergo similar cardiac monitoring as outlined above.
Due to amiodarone's long half-life, patients discontinuing amiodarone just prior to starting EPCLUSA should also undergo similar cardiac monitoring as outlined above.
Patients who develop signs or symptoms of bradycardia should seek medical evaluation immediately. Symptoms may include near-fainting or fainting, dizziness or lightheadedness, malaise, weakness, excessive tiredness, shortness of breath, chest pains, confusion, or memory problems [see Adverse Reactions (6.2) and Drug Interactions (7.3)].
5.3 Risk of Reduced Therapeutic Effect Due to Concomitant Use of EPCLUSA with Inducers of P-gp and/or Moderate to Strong Inducers of CYP
Drugs that are inducers of P-gp and/or moderate to strong inducers of CYP2B6, CYP2C8, or CYP3A4 (e.g., rifampin, St. John's wort, carbamazepine) may significantly decrease plasma concentrations of sofosbuvir and/or velpatasvir, leading to potentially reduced therapeutic effect of EPCLUSA. The use of these agents with EPCLUSA is not recommended [see Drug Interactions (7.3)].
5.4 Risks Associated with Ribavirin and EPCLUSA Combination Treatment
If EPCLUSA is administered with ribavirin, the warnings and precautions for ribavirin apply to this combination regimen. Refer to the ribavirin prescribing information for a full list of the warnings and precautions for ribavirin [see Dosage and Administration (2.2)].
6. Adverse Reactions
The following serious adverse reactions are described below and elsewhere in labeling:
- Serious Symptomatic Bradycardia When Coadministered with Amiodarone [see Warnings and Precautions (5.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
If EPCLUSA is administered with ribavirin, refer to the prescribing information for ribavirin for a description of ribavirin-associated adverse reactions.
Clinical Trials in Adult Subjects
Adverse Reactions in Subjects without Cirrhosis or with Compensated Cirrhosis
The adverse reactions data for EPCLUSA in patients without cirrhosis or with compensated cirrhosis were derived from three Phase 3 clinical trials (ASTRAL-1, ASTRAL-2, and ASTRAL-3) which evaluated a total of 1035 subjects infected with genotype 1, 2, 3, 4, 5, or 6 HCV, without cirrhosis or with compensated cirrhosis, who received EPCLUSA for 12 weeks. EPCLUSA was studied in placebo- and active-controlled trials [see Clinical Studies (14.2)].
The proportion of subjects who permanently discontinued treatment due to adverse events was 0.2% for subjects who received EPCLUSA for 12 weeks.
The most common adverse reactions (adverse events assessed as causally related by the investigator and at least 10%) were headache and fatigue in subjects treated with EPCLUSA for 12 weeks.
Adverse reactions, all grades, observed in greater than or equal to 5% of subjects receiving 12 weeks of treatment with EPCLUSA in ASTRAL-1 include headache (22%), fatigue (15%), nausea (9%), asthenia (5%), and insomnia (5%). Of subjects receiving EPCLUSA who experienced these adverse reactions, 79% had an adverse reaction of mild severity (Grade 1). With the exception of asthenia, each of these adverse reactions occurred at a similar frequency or more frequently in subjects treated with placebo compared to subjects treated with EPCLUSA (asthenia: 3% versus 5% for the placebo and EPCLUSA groups, respectively).
The adverse reactions observed in subjects treated with EPCLUSA in ASTRAL-2 and ASTRAL-3 were consistent with those observed in ASTRAL-1. Irritability was also observed in greater than or equal to 5% of subjects treated with EPCLUSA in ASTRAL-3.
Adverse Reactions in Subjects Coinfected with HCV and HIV-1
The safety assessment of EPCLUSA in subjects with HCV/HIV-1 coinfection was based on an open-label clinical trial (ASTRAL-5) in 106 subjects who were on stable antiretroviral therapy [see Clinical Studies (14.3)]. The safety profile in HCV/HIV-1 coinfected subjects was similar to that observed in HCV mono-infected subjects. The most common adverse reactions occurring in at least 10% of subjects were fatigue (22%) and headache (10%).
Adverse Reactions in Subjects with Decompensated Cirrhosis
The safety assessment of EPCLUSA in subjects infected with genotype 1, 2, 3, 4, or 6 HCV with decompensated cirrhosis was based on one Phase 3 trial (ASTRAL-4) including 87 subjects who received EPCLUSA with ribavirin for 12 weeks. All 87 subjects had Child-Pugh B cirrhosis at screening. On the first day of treatment with EPCLUSA with ribavirin, 6 subjects and 4 subjects were assessed to have Child-Pugh A and Child-Pugh C cirrhosis, respectively [see Clinical Studies (14.4)].
The most common adverse reactions (adverse events assessed as causally related by the investigator, all grades with frequency of 10% or greater) in the 87 subjects who received EPCLUSA with ribavirin for 12 weeks were fatigue (32%), anemia (26%), nausea (15%), headache (11%), insomnia (11%), and diarrhea (10%). Of subjects who experienced these adverse reactions, 98% had adverse reactions of mild to moderate severity.
A total of 4 (5%) subjects permanently discontinued EPCLUSA with ribavirin due to an adverse event; there was no adverse event leading to discontinuation that occurred in more than 1 subject.
Decreases in hemoglobin to less than 10 g/dL and 8.5 g/dL during treatment were observed in 23% and 7% of subjects treated with EPCLUSA with ribavirin for 12 weeks, respectively. Ribavirin was permanently discontinued in 17% of subjects treated with EPCLUSA with ribavirin for 12 weeks, due to adverse reactions.
Less Common Adverse Reactions Reported in Clinical Trials
The following adverse reactions occurred in less than 5% of subjects without cirrhosis or with compensated cirrhosis treated with EPCLUSA for 12 weeks and are included because of a potential causal relationship.
Rash: In the ASTRAL-1 study, rash occurred in 2% of subjects treated with EPCLUSA and in 1% of subjects treated with placebo. No serious adverse reactions of rash occurred, and all rashes were mild or moderate in severity.
Depression: In the ASTRAL-1 study, depressed mood occurred in 1% of subjects treated with EPCLUSA and was not reported by any subject taking placebo. No serious adverse reactions of depressed mood occurred, and all events were mild or moderate in severity.
The following adverse reactions occurred in less than 10% of subjects with decompensated cirrhosis (ASTRAL-4) treated with EPCLUSA with ribavirin for 12 weeks and are included because of a potential causal relationship.
Laboratory Abnormalities
Lipase Elevations: In ASTRAL-1, isolated, asymptomatic lipase elevations of greater than 3×ULN were observed in 3% and 1% of subjects treated with EPCLUSA and placebo for 12 weeks, respectively; and in 6% and 3% of subjects treated with EPCLUSA in ASTRAL-2 and ASTRAL-3, respectively.
In the Phase 3 trial of subjects with decompensated cirrhosis (ASTRAL-4), lipase was assessed when amylase values were greater than or equal to 1.5×ULN. Isolated, asymptomatic lipase elevations of greater than 3×ULN were observed in 2% of subjects treated with EPCLUSA with ribavirin for 12 weeks.
Creatine Kinase: In ASTRAL-1, isolated, asymptomatic creatine kinase elevations greater than or equal to 10×ULN were reported in 1% and 0% of subjects treated with EPCLUSA and placebo for 12 weeks, respectively; and in 2% and 1% of subjects treated with EPCLUSA in ASTRAL-2 and ASTRAL-3, respectively.
In the Phase 3 trial with decompensated cirrhosis (ASTRAL-4), isolated, asymptomatic creatine kinase elevations greater than or equal to 10×ULN were reported in 1% of subjects treated with EPCLUSA with ribavirin for 12 weeks.
Indirect Bilirubin: Increases in indirect bilirubin up to 3 mg/dL above baseline were noted among HIV-1/HCV coinfected subjects treated with EPCLUSA and an atazanavir/ritonavir-based antiretroviral regimen. The elevated indirect bilirubin values were not associated with clinical adverse events, and all subjects completed 12 weeks of EPCLUSA without dose adjustment or treatment interruption of either EPCLUSA or HIV antiretroviral agents.
Adverse Reactions in Adult Liver Transplant Recipients
The safety assessment of EPCLUSA in liver transplant recipients was based on an open-label clinical trial (Trial 2104) in 79 adults without cirrhosis or with compensated cirrhosis who received EPCLUSA for 12 weeks [see Clinical Studies (14.5)]. One subject discontinued treatment due to an adverse event on Day 7. The adverse reactions observed were consistent with the known safety profile of EPCLUSA. Adverse reactions occurring in at least 5% of subjects were headache (18%), fatigue (15%), nausea (8%), diarrhea (6%), and asthenia (5%).
Adverse Reactions in Adults with Severe Renal Impairment Requiring Dialysis
In an open-label trial (Trial 4062), in which a total of 59 adults with HCV with compensated liver disease (with or without cirrhosis) and ESRD requiring dialysis received EPCLUSA for 12 weeks, the most common adverse reaction was nausea (7%) [see Clinical Studies (14.6)].
Adverse Reactions in People Who Inject Drugs (PWID), Including Those on Medication-Assisted Treatment (MAT) for Opioid Use Disorder
The safety of EPCLUSA in PWID is based on an open-label Phase 2 trial (SIMPLIFY) that enrolled 103 adult subjects with chronic HCV genotype 1, 2, 3, and 4 infection. Subjects who self-reported injection drug use within the 6 months prior to starting treatment were eligible and were treated with EPCLUSA for 12 weeks. The trial included a subset of 58 subjects on MAT for opioid use disorder.
The adverse reactions observed from SIMPLIFY both overall and in subjects on MAT were consistent with the known safety profile of EPCLUSA. The most common adverse reactions overall were fatigue (18%), nausea (13%), and headache (11%) [see Use in Specific Populations (8.8) and Clinical Studies (14.7)]. Adverse reactions leading to permanent discontinuation of treatment were not observed in any subjects.
Adverse Reactions in Pediatric Subjects 3 Years of Age and Older
The safety assessment of EPCLUSA in pediatric subjects 3 years of age and older is based on data from a Phase 2, open-label clinical trial (Study 1143) that enrolled 216 subjects who were treated with EPCLUSA for 12 weeks [see Clinical Studies (14.8)]. The adverse reactions observed in pediatric subjects 6 years of age and older were consistent with those observed in clinical trials of EPCLUSA in adults.
Among the 41 pediatric subjects less than 6 years of age, gastrointestinal adverse reactions were reported more commonly compared to subjects 6 years of age and older. Vomiting and product use issue (spitting up the drug) were reported in 15% and 10% of subjects, respectively; these adverse reactions were mild (Grade 1 or 2) and led to treatment discontinuation in 5 (12%) subjects [see Use in Specific Populations (8.4) and Clinical Studies (14.8)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of sofosbuvir. Because postmarketing reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders
Serious symptomatic bradycardia has been reported in patients taking amiodarone who initiate treatment with a sofosbuvir-containing regimen [see Warnings and Precautions (5.2) and Drug Interactions (7.3)].
7. Drug Interactions
7.1 Potential for Other Drugs to Affect EPCLUSA
Sofosbuvir and velpatasvir are substrates of drug transporters P-gp and BCRP while GS-331007 (the predominant circulating metabolite of sofosbuvir) is not. In vitro, slow metabolic turnover of velpatasvir by CYP2B6, CYP2C8, and CYP3A4 was observed.
Drugs that are inducers of P-gp and/or moderate to strong inducers of CYP2B6, CYP2C8, or CYP3A4 (e.g., rifampin, St. John's wort, carbamazepine) may decrease plasma concentrations of sofosbuvir and/or velpatasvir, leading to reduced therapeutic effect of EPCLUSA. The use of these agents with EPCLUSA is not recommended [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)]. EPCLUSA may be coadministered with P-gp, BCRP, and CYP inhibitors.
7.2 Potential for EPCLUSA to Affect Other Drugs
Velpatasvir is an inhibitor of drug transporters P-gp, breast cancer resistance protein (BCRP), OATP1B1, OATP1B3, and OATP2B1. Coadministration of EPCLUSA with drugs that are substrates of these transporters may increase the exposure of such drugs.
7.3 Established and Potentially Significant Drug Interactions
Clearance of HCV infection with direct acting antivirals may lead to changes in hepatic function, which may impact the safe and effective use of concomitant medications. For example, altered blood glucose control resulting in serious symptomatic hypoglycemia has been reported in diabetic patients in postmarketing case reports and published epidemiological studies. Management of hypoglycemia in these cases required either discontinuation or dose modification of concomitant medications used for diabetes treatment.
Frequent monitoring of relevant laboratory parameters (e.g., International Normalized Ratio [INR] in patients taking warfarin, blood glucose levels in diabetic patients) or drug concentrations of concomitant medications such as cytochrome P450 substrates with a narrow therapeutic index (e.g., certain immunosuppressants) is recommended to ensure safe and effective use. Dose adjustments of concomitant medications may be necessary.
Table 4 provides a listing of established or potentially clinically significant drug interactions. The drug interactions described are based on studies conducted with either EPCLUSA, the components of EPCLUSA (sofosbuvir and velpatasvir) as individual agents, or are predicted drug interactions that may occur with EPCLUSA [see Warnings and Precautions (5.2, 5.3) and Clinical Pharmacology (12.3)].
| Concomitant Drug Class: Drug Name | Effect on Concentration† | Clinical Effect/Recommendation |
|---|---|---|
| DF = disoproxil fumarate. | ||
| ||
| Acid Reducing Agents: | ↓ velpatasvir | Velpatasvir solubility decreases as pH increases. Drugs that increase gastric pH are expected to decrease concentration of velpatasvir. |
| Antacids (e.g., aluminum and magnesium hydroxide) | Separate antacid and EPCLUSA administration by 4 hours. | |
| H2-receptor antagonists‡ (e.g., famotidine) | H2-receptor antagonists may be administered simultaneously with or 12 hours apart from EPCLUSA at a dose that does not exceed doses comparable to famotidine 40 mg twice daily. | |
| Proton-pump inhibitors‡ (e.g., omeprazole) | Coadministration of omeprazole or other proton-pump inhibitors is not recommended. If it is considered medically necessary to coadminister, EPCLUSA should be administered with food and taken 4 hours before omeprazole 20 mg. Use with other proton-pump inhibitors has not been studied. | |
| Antiarrhythmics: amiodarone | Effect on amiodarone, sofosbuvir, and velpatasvir concentrations unknown | Coadministration of amiodarone with a sofosbuvir-containing regimen may result in serious symptomatic bradycardia. The mechanism of this effect is unknown. Coadministration of amiodarone with EPCLUSA is not recommended; if coadministration is required, cardiac monitoring is recommended [see Warnings and Precautions (5.2) and Adverse Reactions (6.2)]. |
| digoxin‡ | ↑ digoxin | Therapeutic concentration monitoring of digoxin is recommended when coadministered with EPCLUSA. Refer to digoxin prescribing information for monitoring and dose modification recommendations for concentration increases of less than 50%. |
| Anticancers: topotecan | ↑ topotecan | Coadministration is not recommended. |
| Anticonvulsants: carbamazepine‡ phenytoin phenobarbital | ↓ sofosbuvir ↓ velpatasvir | Coadministration is not recommended. |
| Antimycobacterials: rifabutin‡ rifampin‡ rifapentine | ↓ sofosbuvir ↓ velpatasvir | Coadministration is not recommended. |
| HIV Antiretrovirals: efavirenz‡ | ↓ velpatasvir | Coadministration of EPCLUSA with efavirenz-containing regimens is not recommended. |
| Regimens containing tenofovir DF | ↑ tenofovir | Monitor for tenofovir-associated adverse reactions in patients receiving EPCLUSA concomitantly with a regimen containing tenofovir DF. Refer to the prescribing information of the tenofovir DF-containing product for recommendations on renal monitoring. |
| tipranavir/ritonavir | ↓ sofosbuvir ↓ velpatasvir | Coadministration is not recommended. |
| Herbal Supplements: St. John's wort (Hypericum perforatum) | ↓ sofosbuvir ↓ velpatasvir | Coadministration is not recommended. |
| HMG-CoA Reductase Inhibitors: rosuvastatin‡ | ↑ rosuvastatin | Coadministration of EPCLUSA with rosuvastatin may significantly increase the concentration of rosuvastatin, which is associated with increased risk of myopathy, including rhabdomyolysis. Rosuvastatin may be administered with EPCLUSA at a dose that does not exceed 10 mg. |
| atorvastatin‡ | ↑ atorvastatin | Coadministration of EPCLUSA with atorvastatin may be associated with increased risk of myopathy, including rhabdomyolysis. Monitor closely for HMG-CoA reductase inhibitor-associated adverse reactions, such as myopathy and rhabdomyolysis. |
7.4 Drugs without Clinically Significant Interactions with EPCLUSA
Based on drug interaction studies conducted with the components of EPCLUSA (sofosbuvir or velpatasvir) or EPCLUSA, no clinically significant drug interactions have been observed or are expected with the following drugs [see Clinical Pharmacology (12.3)]:
- EPCLUSA: atazanavir/ritonavir, buprenorphine/naloxone, cyclosporine, darunavir/ritonavir, dolutegravir, elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide, emtricitabine, methadone, naltrexone, raltegravir, or rilpivirine.
- Sofosbuvir: ethinyl estradiol/norgestimate, or tacrolimus.
- Velpatasvir: ethinyl estradiol/norgestimate, ketoconazole, or pravastatin.
See Table 4 for use of EPCLUSA with certain HIV antiretroviral regimens [see Drug Interactions (7.3)].
8. Use in Specific Populations
8.1 Pregnancy
Risk Summary
If EPCLUSA is administered with ribavirin, the combination regimen is contraindicated in pregnant women and in men whose female partners are pregnant. Refer to the ribavirin prescribing information for more information on ribavirin-associated risks of use during pregnancy.
No adequate human data are available to establish whether or not EPCLUSA poses a risk to pregnancy outcomes. In animal reproduction studies, no evidence of adverse developmental outcomes was observed with the components of EPCLUSA (sofosbuvir or velpatasvir) at exposures greater than those in humans at the recommended human dose (RHD) [see Data]. During organogenesis in the mouse, rat, and rabbit, systemic exposures (AUC) to velpatasvir were approximately 31 (mice), 6 (rats), and 0.4 (rabbits) times the exposure in humans at the RHD, while exposures to the predominant circulating metabolite of sofosbuvir (GS-331007) were approximately 4 (rats) and 10 (rabbits) times the exposure in humans at the RHD. In rat pre/postnatal development studies, maternal systemic exposures (AUC) to velpatasvir and GS-331007 were approximately 5 times the exposures of each component in humans at the RHD.
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Data
Sofosbuvir: Sofosbuvir was administered orally to pregnant rats (up to 500 mg/kg/day) and rabbits (up to 300 mg/kg/day) on gestation days 6 to 18 and 6 to 19, respectively, and also to rats (oral doses up to 500 mg/kg/day) on gestation day 6 to lactation/post-partum day 20. No significant effects on embryo-fetal (rats and rabbits) or pre/postnatal (rats) development were observed at the highest doses tested. The systemic exposures (AUC) of the predominant circulating metabolite of sofosbuvir (GS-331007) during gestation were approximately 4 (rats) and 10 (rabbits) times the exposure in humans at the RHD.
Velpatasvir: Velpatasvir was administered orally to pregnant mice (up to 1000 mg/kg/day), rats (up to 200 mg/kg/day), and rabbits (up to 300 mg/kg/day) on gestation days 6 to 15, 6 to 17, and 7 to 20, respectively, and also to rats (oral doses up to 200 mg/kg) on gestation day 6 to lactation/post-partum day 20. No significant effects on embryo-fetal (mice, rats, and rabbits) or pre/postnatal (rats) development were observed at the highest doses tested. The systemic exposures (AUC) of velpatasvir during gestation were approximately 31 (mice), 6 (rats), and 0.4 (rabbits) times the exposure in humans at the RHD.
8.2 Lactation
Risk Summary
It is not known whether the components of EPCLUSA and its metabolites are present in human breast milk, affect human milk production, or have effects on the breastfed infant. The predominant circulating metabolite of sofosbuvir (GS-331007) was the primary component observed in the milk of lactating rats administered sofosbuvir, without effect on nursing pups. When administered to lactating rats, velpatasvir was detected in the milk of lactating rats and in the plasma of nursing pups without effects on the nursing pups [see Data].
The development and health benefits of breastfeeding should be considered along with the mother's clinical need for EPCLUSA and any potential adverse effects on the breastfed child from EPCLUSA or from the underlying maternal condition.
If EPCLUSA is administered with ribavirin, the nursing mother's information for ribavirin also applies to this combination regimen. Refer to the ribavirin prescribing information for more information on use during lactation.
Data
Sofosbuvir: No effects of sofosbuvir on growth and postnatal development were observed in nursing pups at the highest dose tested in rats. Maternal systemic exposure (AUC) to the predominant circulating metabolite of sofosbuvir (GS-331007) was approximately 5 times the exposure in humans at the RHD, with exposure of approximately 2% that of maternal exposure observed in nursing pups on lactation day 10. In a lactation study, sofosbuvir metabolites (primarily GS-331007) were excreted into the milk of lactating rats following administration of a single oral dose of sofosbuvir (20 mg/kg) on lactation day 2, with milk concentrations of approximately 10% that of maternal plasma concentrations observed 1-hour post-dose.
Velpatasvir: No effects of velpatasvir on growth and postnatal development were observed in nursing pups at the highest dose tested in rats. Maternal systemic exposure (AUC) to velpatasvir was approximately 5 times the exposure in humans at the RHD. Velpatasvir was present in the milk (approximately 173% that of maternal plasma concentrations) of lactating rats following a single oral dose of velpatasvir (30 mg/kg), and systemic exposure (AUC) in nursing pups was approximately 4% that of maternal exposure on lactation day 10.
8.3 Females and Males of Reproductive Potential
If EPCLUSA is administered with ribavirin, the information for ribavirin with regard to pregnancy testing, contraception, and infertility also applies to this combination regimen. Refer to ribavirin prescribing information for additional information.
8.4 Pediatric Use
The pharmacokinetics, safety, and effectiveness of EPCLUSA for treatment of HCV genotype 1, 2, 3, 4, or 6 infection in treatment-naïve and treatment-experienced pediatric patients 3 years of age and older without cirrhosis or with compensated cirrhosis have been established in an open-label, multicenter clinical trial (Study 1143, N=216; 190 treatment-naïve, 26 treatment-experienced). No clinically meaningful differences in pharmacokinetics were observed in comparison to those observed in adults.
The safety and effectiveness in pediatric subjects were comparable to those observed in adults. However, among the 41 pediatric subjects less than 6 years of age, vomiting and product use issue (spitting up the drug) were reported more frequently (15% and 10%, respectively; all Grade 1 or 2) compared to subjects 6 years of age and older. Five subjects (12%) discontinued treatment after vomiting or spitting up the drug [see Dosage and Administration (2.4, 2.5), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.8)].
The safety and effectiveness of EPCLUSA for treatment of HCV genotype 5 in pediatric patients 3 years of age and older without cirrhosis or with compensated cirrhosis are supported by sofosbuvir, GS-331007, and velpatasvir exposures in adults and pediatric patients [see Dosage and Administration (2.2 and 2.4), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.8)]. Similar rationale is used to support dosing recommendations for pediatric patients with HCV genotype 1, 2, 3, 4, 5, or 6 infection who have decompensated cirrhosis (Child-Pugh B or C).
In patients with severe renal impairment, including those requiring dialysis, exposures of GS-331007, the inactive metabolite of sofosbuvir, are increased [see Clinical Pharmacology (12.3)]. No data are available regarding the safety of EPCLUSA in pediatric patients with renal impairment [see Use in Specific Populations (8.6)].
The safety and effectiveness of EPCLUSA have not been established in pediatric patients less than 3 years of age.
8.5 Geriatric Use
Clinical trials of EPCLUSA included 156 subjects aged 65 and over (12% of total number of subjects in the Phase 3 clinical trials). No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. No dosage adjustment of EPCLUSA is warranted in geriatric patients [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
No dosage adjustment of EPCLUSA is recommended for patients with mild, moderate, or severe renal impairment, including ESRD requiring dialysis [see Dosage and Administration (2.6), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.6)]. No safety data are available in subjects with both decompensated cirrhosis and severe renal impairment, including ESRD requiring dialysis. Additionally, no safety data are available in pediatric patients with renal impairment [see Use in Specific Populations (8.4)]. Refer to ribavirin tablet prescribing information regarding use of ribavirin in patients with renal impairment.
8.7 Hepatic Impairment
No dosage adjustment of EPCLUSA is recommended for patients with mild, moderate, or severe hepatic impairment (Child-Pugh Class A, B, or C) [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)].
Clinical and hepatic laboratory monitoring (including direct bilirubin), as clinically indicated, is recommended for patients with decompensated cirrhosis receiving treatment with EPCLUSA and ribavirin [see Adverse Reactions (6.1)].
8.8 People Who Inject Drugs (PWID), Including Those on Medication-Assisted Treatment (MAT) for Opioid Use Disorder
Based on data from the Phase 2 trial SIMPLIFY, the safety and effectiveness of EPCLUSA in subjects who self-reported injection drug use, including in those on concomitant MAT, were similar to the known safety and effectiveness profile of EPCLUSA. No dosage adjustment of EPCLUSA is recommended for PWID, including those on MAT for opioid use disorder [see Adverse Reactions (6.1) and Clinical Studies (14.7)].
10. Overdosage
No specific antidote is available for overdose with EPCLUSA. If overdose occurs the patient must be monitored for evidence of toxicity. Treatment of overdose with EPCLUSA consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the patient. Hemodialysis can efficiently remove the predominant circulating metabolite of sofosbuvir, GS-331007, with an extraction ratio of 53%. Hemodialysis is unlikely to result in significant removal of velpatasvir since velpatasvir is highly bound to plasma protein.
11. Description
Tablets
EPCLUSA is a fixed-dose combination tablet containing sofosbuvir and velpatasvir for oral administration. Sofosbuvir is a nucleotide analog HCV NS5B polymerase inhibitor and velpatasvir is an NS5A inhibitor.
Each 400 mg/100 mg tablet contains 400 mg sofosbuvir and 100 mg velpatasvir, and each 200 mg/50 mg tablet contains 200 mg sofosbuvir and 50 mg velpatasvir. The tablets include the following inactive ingredients: copovidone, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing the following inactive ingredients: iron oxide red, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
Pellets
EPCLUSA oral pellets are for oral administration, supplied as small, white to off-white pellets in unit-dose packets. Each unit-dose of EPCLUSA oral pellets contains either 200 mg sofosbuvir and 50 mg velpatasvir or 150 mg sofosbuvir and 37.5 mg velpatasvir and the following inactive ingredients: colloidal silicon dioxide, copovidone, croscarmellose sodium, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. The oral pellets are film-coated with a coating material containing the following inactive ingredients: amino methacrylate copolymer, colloidal silicon dioxide, hypromellose, L-tartaric acid, polyethylene glycol, stearic acid, talc, and titanium dioxide.
Sofosbuvir: The IUPAC name for sofosbuvir is (S)-isopropyl 2-((S)-(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)-(phenoxy)phosphorylamino)propanoate. It has a molecular formula of C22H29FN3O9P and a molecular weight of 529.45. It has the following structural formula:
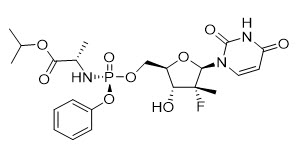
Sofosbuvir is a white to off-white crystalline solid with a solubility of at least 2 mg/mL across the pH range of 2–7.7 at 37 °C and is slightly soluble in water.
Velpatasvir: The IUPAC name for velpatasvir is methyl {(1R)-2-[(2S,4S)-2-(5-{2-[(2S,5S)-1-{(2S)-2-[(methoxycarbonyl)amino]-3-methylbutanoyl}-5-methylpyrrolidin-2-yl]-1,11-dihydro[2]benzopyrano[4',3':6,7]naphtho[1,2-d]imidazol-9-yl}-1H-imidazol-2-yl)-4-(methoxymethyl)pyrrolidin-1-yl]-2-oxo-1-phenylethyl}carbamate. It has a molecular formula of C49H54N8O8 and a molecular weight of 883.0. It has the following structural formula:
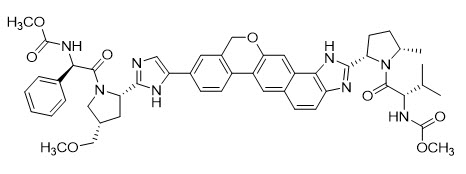
Velpatasvir is practically insoluble (less than 0.1 mg/mL) above pH 5, slightly soluble (3.6 mg/mL) at pH 2, and soluble (greater than 36 mg/mL) at pH 1.2.
12. Clinical Pharmacology
12.1 Mechanism of Action
EPCLUSA is a fixed-dose combination of sofosbuvir and velpatasvir, which are direct-acting antiviral agents against the hepatitis C virus [see Microbiology (12.4)].
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of sofosbuvir 400 mg (recommended dosage) and 1200 mg (3 times the recommended dosage) on QTc interval was evaluated in an active-controlled (moxifloxacin 400 mg) thorough QT trial. At a dose 3 times the recommended dose, sofosbuvir does not prolong QTc to any clinically relevant extent.
The effect of velpatasvir 500 mg (5 times the recommended dosage) was evaluated in an active-controlled (moxifloxacin 400 mg) thorough QT trial. At a dose 5 times the recommended dose, velpatasvir does not prolong QTc interval to any clinically relevant extent.
12.3 Pharmacokinetics
The pharmacokinetic properties of the components of EPCLUSA are provided in Table 5. The multiple dose pharmacokinetic parameters of sofosbuvir and its metabolite, GS-331007, and velpatasvir are provided in Table 6.
| Sofosbuvir | Velpatasvir | |
|---|---|---|
| CES1 = carboxylesterase 1; HINT1 = histidine triad nucleotide-binding protein 1. | ||
| ||
| Absorption | ||
| Tmax (hr) | 0.5–1 | 3 |
| Effect of moderate meal (relative to fasting)* | ↑ 60% | ↑ 34% |
| Effect of high fat meal (relative to fasting)* | ↑ 78% | ↑ 21% |
| Distribution | ||
| % Bound to human plasma proteins | 61–65 | >99.5 |
| Blood-to-plasma ratio | 0.7 | 0.52–0.67 |
| Metabolism | ||
| Metabolism | Cathepsin A CES1 HINT1 | CYP2B6 CYP2C8 CYP3A4 |
| Elimination | ||
| Major route of elimination | SOF: metabolism GS-331007†: glomerular filtration and active tubular secretion | Biliary excretion as parent (77%) |
| t1/2 (hr)‡ | SOF: 0.5 GS-331007†: 25 | 15 |
| % Of dose excreted in urine§ | 80¶ | 0.4 |
| % Of dose excreted in feces§ | 14 | 94 |
| Parameter Mean (%CV) | Sofosbuvir* | GS-331007† | Velpatasvir‡ |
|---|---|---|---|
| CV = coefficient of variation; NA = not applicable. | |||
| |||
| Cmax
(ng/mL) | 567 (30.7) | 898 (26.7) | 259 (54.3) |
| AUCtau
(ng∙hr/mL) | 1268 (38.5) | 14372 (28.0) | 2980 (51.3) |
| Ctrough
(ng/mL) | NA | - | 42 (67.3) |
Sofosbuvir and GS-331007 AUC0–24 and Cmax were similar in healthy adult subjects and subjects with HCV infection. Relative to healthy subjects (N=331), velpatasvir AUC0–24 and Cmax were 37% lower and 42% lower, respectively, in HCV-infected subjects.
Velpatasvir AUC increases in a greater than proportional manner from 5 to 50 mg and in a less than proportional manner from 50 to 450 mg in healthy volunteers. However, velpatasvir exhibited more than or near dose-proportional increase in exposures 25 mg to 150 mg in HCV-infected patients when coadministered with sofosbuvir. Sofosbuvir and GS-331007 AUCs are near dose-proportional over the dose range of 200 mg to 1200 mg.
Specific Populations
Pediatric Patients: The pharmacokinetics of sofosbuvir, GS-331007, and velpatasvir were determined in HCV genotype 1, 2, 3, 4, or 6 infected pediatric subjects 3 years of age and older receiving a daily dose of EPCLUSA as described below in Table 7. Sofosbuvir AUCtau and Cmax and velpatasvir Cmax values were 67%, 69%, and 78% higher in pediatric subjects ≥30 kg, 68%, 70%, and 96% higher in pediatric subjects 17 to <30 kg, and 103%, 135%, and 92% higher in pediatric subjects <17 kg compared to those observed in adults. These differences were not considered clinically significant. GS-331007 exposures and velpatasvir AUCtau and Ctau values in pediatric subjects were similar to those observed in adults.
| Weight Group | Dose | PK Parameter | Mean (%CV) | |||
|---|---|---|---|---|---|---|
| Sofosbuvir | GS-331007 | Velpatasvir | ||||
| CV = coefficient of variation; NA = not applicable. | ||||||
| ||||||
| <17 kg† | 150/37.5 mg | Cmax (ng/mL) | 1550 (65.2) | 1090 (17.0) | 488 (46.6) | |
| AUCtau (ng∙hr/mL) | 2830 (63.7) | 11900 (19.7) | 4480 (53.4) | |||
| Ctrough (ng/mL) | NA | - | 57.4 (82.7) | |||
| 17 to <30 kg‡ | 200/50 mg | Cmax (ng/mL) | 1200 (73.8) | 1070 (27.2) | 483 (39.5) | |
| AUCtau (ng∙hr/mL) | 2280 (55.6) | 11400 (43.3) | 4090 (38.5) | |||
| Ctrough (ng/mL) | NA | - | 43 (65.8) | |||
| ≥30 kg§ | 400/100 mg | Cmax (ng/mL) | 1310 (91.4) | 1180 (24.6) | 456 (56.4) | |
| AUCtau (ng∙hr/mL) | 2570 (82.8) | 13600 (27.6) | 4240 (46.7) | |||
| Ctrough (ng/mL) | NA | - | 42.2 (66.4) | |||
The pharmacokinetics of sofosbuvir, GS-331007 and velpatasvir have not been established in pediatric subjects less than 3 years of age [see Use in Specific Populations (8.4) and Clinical Studies (14.7)].
Geriatric Patients: Population pharmacokinetic analysis in HCV-infected subjects showed that within the age range (18 to 82 years) analyzed, age did not have a clinically relevant effect on the exposure to sofosbuvir, GS-331007, or velpatasvir [see Use in Specific Populations (8.5)].
Patients with Renal Impairment:
The pharmacokinetics of sofosbuvir were studied in HCV negative subjects with mild (eGFR between 50 to less than 80 mL/min/1.73 m2), moderate (eGFR between 30 to less than 50 mL/min/1.73 m2), severe renal impairment (eGFR less than 30 mL/min/1.73 m2), and subjects with ESRD requiring hemodialysis following a single 400 mg dose of sofosbuvir. Relative to subjects with normal renal function (eGFR greater than 80 mL/min/1.73 m2), the sofosbuvir AUC0–inf was 61%, 107%, and 171% higher in subjects with mild, moderate, and severe renal impairment, while the GS-331007 AUC0–inf was 55%, 88%, and 451% higher, respectively.
In subjects with ESRD, relative to subjects with normal renal function, sofosbuvir and GS-331007 AUC0–inf was 28% and 1280% higher when sofosbuvir was dosed 1 hour before hemodialysis compared with 60% and 2070% higher when sofosbuvir was dosed 1 hour after hemodialysis, respectively. A 4-hour hemodialysis session removed approximately 18% of administered dose [see Dosage and Administration (2.6) and Use in Specific Populations (8.6)].
The pharmacokinetics of velpatasvir were studied with a single dose of 100 mg velpatasvir in HCV negative subjects with severe renal impairment (eGFR less than 30 mL/min by Cockcroft-Gault). No clinically relevant differences in velpatasvir pharmacokinetics were observed between healthy subjects and subjects with severe renal impairment [see Use in Specific Populations (8.6)].
The pharmacokinetics of sofosbuvir, GS-331007, and velpatasvir were studied in HCV-infected subjects with ESRD requiring dialysis treated with EPCLUSA for 12 weeks. The results were generally consistent with those in HCV negative subjects with ESRD requiring dialysis.
Patients with Hepatic Impairment:
The pharmacokinetics of sofosbuvir were studied following 7-day dosing of 400 mg sofosbuvir in HCV-infected subjects with moderate and severe hepatic impairment (Child-Pugh Class B and C, respectively). Relative to subjects with normal hepatic function, the sofosbuvir AUC0–24 were 126% and 143% higher in moderate and severe hepatic impairment, while the GS-331007 AUC0–24 were 18% and 9% higher, respectively. Population pharmacokinetics analysis in HCV-infected subjects indicated that cirrhosis (including decompensated cirrhosis) had no clinically relevant effect on the exposure of sofosbuvir and GS-331007 [see Use in Specific Populations (8.7)].
The pharmacokinetics of velpatasvir were studied with a single dose of 100 mg velpatasvir in HCV negative subjects with moderate and severe hepatic impairment (Child-Pugh Class B and C). Velpatasvir plasma exposure (AUCinf) was similar in subjects with moderate hepatic impairment, severe hepatic impairment, and control subjects with normal hepatic function. Population pharmacokinetics analysis in HCV-infected subjects indicated that cirrhosis (including decompensated cirrhosis) had no clinically relevant effect on the exposure of velpatasvir [see Use in Specific Populations (8.7)].
Drug Interaction Studies
After oral administration of EPCLUSA, sofosbuvir is rapidly absorbed and subject to extensive first-pass hepatic extraction (hydrolysis followed by sequential phosphorylation) to form the pharmacologically active triphosphate. In clinical pharmacology studies, both sofosbuvir and the primary circulating metabolite GS-331007 (dephosphorylated nucleotide metabolite) were monitored for purposes of pharmacokinetic analyses.
Sofosbuvir and velpatasvir are substrates of drug transporters P-gp and BCRP while GS-331007 is not. Velpatasvir is also transported by OATP1B1 and OATP1B3. In vitro, slow metabolic turnover of velpatasvir by CYP2B6, CYP2C8, and CYP3A4 was observed. Inducers of P-gp and/or moderate to strong inducers of CYP2B6, CYP2C8, or CYP3A4 (e.g., rifampin, St. John's wort, carbamazepine) may decrease plasma concentrations of sofosbuvir and/or velpatasvir, leading to reduced therapeutic effect of EPCLUSA [see Warnings and Precautions (5.3) and Drug Interactions (7.3)]. Coadministration with drugs that inhibit P-gp and/or BCRP may increase sofosbuvir and/or velpatasvir plasma concentrations without increasing GS-331007 plasma concentration. Drugs that inhibit CYP2B6, CYP2C8, or CYP3A4 may increase plasma concentration of velpatasvir.
Velpatasvir is an inhibitor of drug transporter P-gp, BCRP, OATP1B1, OATP1B3, and OATP2B1, and its involvement in drug interactions with these transporters is primarily limited to the process of absorption. At clinically relevant concentration, velpatasvir is not an inhibitor of hepatic transporters OATP1A2 or OCT1, renal transporters OCT2, OAT1, OAT3, or MATE1, or CYP or UGT1A1 enzymes.
Sofosbuvir and GS-331007 are not inhibitors of drug transporters P-gp, BCRP, OATP1B1, OATP1B3, and OCT1 and GS-331007 is not an inhibitor of OAT1, OAT3, OCT2, and MATE1. Sofosbuvir and GS-331007 are not inhibitors or inducers of CYP or UGT1A1 enzymes.
The effects of coadministered drugs on the exposure of sofosbuvir, GS-331007, and velpatasvir are shown in Table 8. The effects of sofosbuvir, velpatasvir, or EPCLUSA on the exposure of coadministered drugs are shown in Table 9 [see Drug Interactions (7)].
| Coadministered Drug | Dose of Coadministered Drug (mg) | SOF Dose (mg) | VEL Dose (mg) | N | Mean Ratio (90% CI) of Sofosbuvir, GS-331007, and Velpatasvir PK With/Without Coadministered Drug No Effect=1.00 | ||||
|---|---|---|---|---|---|---|---|---|---|
| Cmax | AUC | Cmin | |||||||
| NA = not available/not applicable, ND = not dosed, DF = disoproxil fumarate. | |||||||||
| |||||||||
| Atazanavir/ ritonavir + emtricitabine/ tenofovir DF | 300/100 + 200/300 once daily | 400 once daily | 100 once daily | 24 | sofosbuvir | 1.12 (0.97, 1.29) | 1.22 (1.12, 1.33) | NA | |
| GS-331007 | 1.21 (1.12, 1.29) | 1.32 (1.27, 1.36) | 1.42 (1.37, 1.49) | ||||||
| velpatasvir | 1.55 (1.41, 1.71) | 2.42 (2.23, 2.64) | 4.01 (3.57, 4.50) | ||||||
| Carbamazepine | 300 twice daily | 400 single dose | ND | 24 | sofosbuvir | 0.52 (0.43, 0.62) | 0.52 (0.46, 0.59) | NA | |
| GS-331007 | 1.04 (0.97, 1.11) | 0.99 (0.94, 1.04) | NA | ||||||
| Cyclosporine | 600 single dose | 400 single dose | ND | 19 | sofosbuvir | 2.54 (1.87, 3.45) | 4.53 (3.26, 6.30) | NA | |
| GS-331007 | 0.60 (0.53, 0.69) | 1.04 (0.90, 1.20) | NA | ||||||
| ND | 100 single dose | 12 | velpatasvir | 1.56 (1.22, 2.01) | 2.03 (1.51, 2.71) | NA | |||
| Darunavir/ ritonavir + emtricitabine/ tenofovir DF | 800/100 + 200/300 once daily | 400 once daily | 100 once daily | 29 | sofosbuvir | 0.62 (0.54, 0.71) | 0.72 (0.66, 0.80) | NA | |
| GS-331007 | 1.04 (0.99, 1.08) | 1.13 (1.08, 1.18) | 1.13 (1.06, 1.19) | ||||||
| velpatasvir | 0.76 (0.65, 0.89) | 0.84 (0.72, 0.98) | 1.01 (0.87, 1.18) | ||||||
| Efavirenz/ emtricitabine/ tenofovir DF† | 600/200/300 once daily | 400 once daily | 100 once daily | 14 | sofosbuvir | 1.38 (1.14, 1.67) | 0.97 (0.83, 1.14) | NA | |
| GS-331007 | 0.86 (0.80, 0.93) | 0.90 (0.85, 0.96) | 1.01 (0.95, 1.07) | ||||||
| velpatasvir | 0.53 (0.43, 0.64) | 0.47 (0.39, 0.57) | 0.43 (0.36, 0.52) | ||||||
| Elvitegravir/ cobicistat/emtricitabine/tenofovir alafenamide‡ | 150/150/200/10 once daily | 400 once daily | 100 once daily | 24 | sofosbuvir | 1.23 (1.07, 1.42) | 1.37 (1.24, 1.52) | NA | |
| GS-331007 | 1.29 (1.25, 1.33) | 1.48 (1.43, 1.53) | 1.58 (1.52, 1.65) | ||||||
| velpatasvir | 1.30 (1.17, 1.45) | 1.50 (1.35, 1.66) | 1.60 (1.44, 1.78) | ||||||
| Elvitegravir/ cobicistat/emtricitabine/tenofovir DF§ | 150/150/200/300 once daily | 400 once daily | 100 once daily | 24 | sofosbuvir | 1.01 (0.85, 1.19) | 1.24 (1.13, 1.37) | NA | |
| GS-331007 | 1.13 (1.07, 1.18) | 1.35 (1.30, 1.40) | 1.45 (1.38, 1.52) | ||||||
| velpatasvir | 1.05 (0.93, 1.19) | 1.19 (1.07, 1.34) | 1.37 (1.22, 1.54) | ||||||
| Famotidine | 40 single dose simultaneously with EPCLUSA | 400 single dose | 100 single dose | 60 | sofosbuvir | 0.92 (0.82, 1.05) | 0.82 (0.74, 0.91) | NA | |
| GS-331007 | 0.84 (0.78, 0.89) | 0.94 (0.91, 0.98) | NA | ||||||
| velpatasvir | 0.80 (0.70, 0.91) | 0.81 (0.71, 0.91) | NA | ||||||
| 40 single dose 12 hours prior to EPCLUSA | 60 | sofosbuvir | 0.77 (0.68, 0.87) | 0.80 (0.73, 0.88) | NA | ||||
| GS-331007 | 1.20 (1.13, 1.28) | 1.04 (1.01, 1.08) | NA | ||||||
| velpatasvir | 0.87 (0.76, 1.00) | 0.85 (0.74, 0.97) | NA | ||||||
| Ketoconazole | 200 twice daily | ND | 100 single dose | 12 | velpatasvir | 1.29 (1.02, 1.64) | 1.71 (1.35, 2.18) | NA | |
| Lopinavir/ ritonavir + emtricitabine/ tenofovir DF | 4×200/50 + 200/300 once daily | 400 once daily | 100 once daily | 24 | sofosbuvir | 0.59 (0.49, 0.71) | 0.71 (0.64, 0.78) | NA | |
| GS-331007 | 1.01 (0.98, 1.05) | 1.15 (1.09, 1.21) | 1.15 (1.07, 1.25) | ||||||
| velpatasvir | 0.70 (0.59, 0.83) | 1.02 (0.89, 1.17) | 1.63 (1.43, 1.85) | ||||||
| Methadone | 30 to 130 daily | 400 once daily | ND | 14 | sofosbuvir | 0.95 (0.68, 1.33) | 1.30 (1.00, 1.69) | NA | |
| GS-331007 | 0.73 (0.65, 0.83) | 1.04 (0.89, 1.22) | NA | ||||||
| Omeprazole | 20 once daily simultaneously with EPCLUSA | 400 single dose fasted | 100 single dose fasted | 60 | sofosbuvir | 0.66 (0.55, 0.78) | 0.71 (0.60, 0.83) | NA | |
| GS-331007 | 1.18 (1.10, 1.26) | 1.00 (0.95, 1.05) | NA | ||||||
| velpatasvir | 0.63 (0.50, 0.78) | 0.64 (0.52, 0.79) | NA | ||||||
| 20 once daily 12 hours prior to EPCLUSA | 400 single dose fasted | 100 single dose fasted | 60 | sofosbuvir | 0.55 (0.47, 0.64) | 0.56 (0.49, 0.65) | NA | ||
| GS-331007 | 1.26 (1.18, 1.34) | 0.97 (0.94, 1.01) | NA | ||||||
| velpatasvir | 0.43 (0.35, 0.54) | 0.45 (0.37, 0.55) | NA | ||||||
| 20 once daily 2 hours prior to EPCLUSA | 400 single dose fed¶ | 100 single dose fed¶ | 40 | sofosbuvir | 0.84 (0.68, 1.03) | 1.08 (0.94, 1.25) | NA | ||
| GS-331007 | 0.94 (0.88, 1.02) | 0.99 (0.96, 1.03) | NA | ||||||
| velpatasvir | 0.52 (0.43, 0.64) | 0.62 (0.51, 0.75) | NA | ||||||
| 20 once daily 4 hours after EPCLUSA | 400 single dose fed¶ | 100 single dose fed¶ | 38 | sofosbuvir | 0.79 (0.68, 0.92) | 1.05 (0.94, 1.16) | NA | ||
| GS-331007 | 0.91 (0.85, 0.98) | 0.99 (0.95, 1.02) | NA | ||||||
| velpatasvir | 0.67 (0.58, 0.78) | 0.74 (0.63, 0.86) | NA | ||||||
| 40 once daily 4 hours after EPCLUSA | 400 single dose fed¶ | 100 single dose fed¶ | 40 | sofosbuvir | 0.70 (0.57, 0.87) | 0.91 (0.76, 1.08) | NA | ||
| GS-331007 | 1.01 (0.96, 1.07) | 0.99 (0.94, 1.03) | NA | ||||||
| velpatasvir | 0.44 (0.34, 0.57) | 0.47 (0.37, 0.60) | NA | ||||||
| Rifabutin | 300 once daily | 400 single dose | ND | 20 | sofosbuvir | 0.64 (0.53, 0.77) | 0.76 (0.63, 0.91) | NA | |
| GS-331007 | 1.15 (1.03, 1.27) | 1.03 (0.95, 1.12) | NA | ||||||
| Rifampin | 600 once daily | 400 single dose | ND | 17 | sofosbuvir | 0.23 (0.19, 0.29) | 0.28 (0.24, 0.32) | NA | |
| GS-331007 | 1.23 (1.14, 1.34) | 0.95 (0.88, 1.03) | NA | ||||||
| ND | 100 single dose | 12 | velpatasvir | 0.29 (0.23, 0.37) | 0.18 (0.15, 0.22) | NA | |||
| 600 single dose | ND | 100 single dose | 12 | velpatasvir | 1.28 (1.05, 1.56) | 1.46 (1.17, 1.83) | NA | ||
| Tacrolimus | 5 single dose | 400 single dose | ND | 16 | sofosbuvir | 0.97 (0.65, 1.43) | 1.13 (0.81, 1.57) | NA | |
| GS-331007 | 0.97 (0.83, 1.14) | 1.00 (0.87, 1.13) | NA | ||||||
No effect on the pharmacokinetic parameters of sofosbuvir, GS-331007, or velpatasvir was observed with dolutegravir; the combination of emtricitabine, rilpivirine, and tenofovir DF; emtricitabine; raltegravir; or tenofovir DF.
| Coadministered Drug | Dose of Coadministered Drug (mg) | SOF Dose (mg) | VEL Dose (mg) | N | Mean Ratio (90% CI) of Coadministered Drug PK With/Without Sofosbuvir, Velpatasvir, or EPCLUSA No Effect=1.00 | |||
|---|---|---|---|---|---|---|---|---|
| Cmax | AUC | Cmin | ||||||
| NA = not available/not applicable, ND = not dosed, DF = disoproxil fumarate. | ||||||||
| ||||||||
| Atazanavir/ ritonavir + emtricitabine/ tenofovir DF† | atazanavir 300 once daily | 400 once daily | 100 once daily | 24 | 1.09 (1.00, 1.19) | 1.20 (1.10, 1.31) | 1.39 (1.20, 1.61) | |
| ritonavir 100 once daily | 0.89 (0.82, 0.97) | 0.97 (0.89, 1.05) | 1.29 (1.15, 1.44) | |||||
| emtricitabine 200 once daily | 1.01 (0.96, 1.06) | 1.02 (0.99, 1.04) | 1.06 (1.02, 1.11) | |||||
| tenofovir DF 300 once daily | 1.55 (1.43, 1.68) | 1.30 (1.24, 1.36) | 1.39 (1.31, 1.48) | |||||
| Atorvastatin | 40 single dose | 400 once daily | 100 once daily | 26 | 1.68 (1.49, 1.89) | 1.54 (1.45, 1.64) | NA | |
| Darunavir/ ritonavir + emtricitabine/ tenofovir DF‡ | darunavir 800 once daily | 400 once daily | 100 once daily | 29 | 0.90 (0.86, 0.95) | 0.92 (0.87, 0.98) | 0.87 (0.79, 0.95) | |
| ritonavir 100 once daily | 1.07 (0.97, 1.17) | 1.12 (1.05, 1.19) | 1.09 (1.02, 1.15) | |||||
| emtricitabine 200 once daily | 1.05 (1.01, 1.08) | 1.05 (1.02, 1.08) | 1.04 (0.98, 1.09) | |||||
| tenofovir DF 300 once daily | 1.55 (1.45, 1.66) | 1.39 (1.33, 1.44) | 1.52 (1.45, 1.59) | |||||
| Digoxin | 0.25 single dose | ND | 100 | 21 | 1.88 (1.71, 2.08) | 1.34 (1.13, 1.60) | NA | |
| Efavirenz/ emtricitabine/ tenofovir DF§ | efavirenz 600 once daily | 400 once daily | 100 once daily | 15 | 0.81 (0.74, 0.89) | 0.85 (0.80, 0.91) | 0.90 (0.85, 0.95) | |
| emtricitabine 200 once daily | 1.07 (0.98, 1.18) | 1.07 (1.00, 1.14) | 1.10 (0.97, 1.25) | |||||
| tenofovir DF 300 once daily | 1.77 (1.53, 2.04) | 1.81 (1.68, 1.94) | 2.21 (2.00, 2.43) | |||||
| Elvitegravir/ cobicistat/emtricitabine/tenofovir alafenamide¶ | elvitegravir 150 once daily | 400 once daily | 100 once daily | 24 | 0.87 (0.80, 0.94) | 0.94 (0.88, 1.00) | 1.08 (0.97, 1.20) | |
| cobicistat 150 once daily | 1.16 (1.09, 1.23) | 1.30 (1.23, 1.38) | 2.03 (1.67, 2.48) | |||||
| emtricitabine 200 once daily | 1.02 (0.97, 1.06) | 1.01 (0.98, 1.04) | 1.02 (0.97, 1.07) | |||||
| tenofovir alafenamide 10 once daily | 0.80 (0.68, 0.94) | 0.87 (0.81, 0.94) | NA | |||||
| Elvitegravir/ cobicistat/emtricitabine/tenofovir DF# | elvitegravir 150 once daily | 400 once daily | 100 once daily | 24 | 0.93 (0.86, 1.00) | 0.93 (0.87, 0.99) | 0.97 (0.91, 1.04) | |
| cobicistat 150 once daily | 1.11 (1.06, 1.17) | 1.23 (1.17, 1.29) | 1.71 (1.54, 1.90) | |||||
| emtricitabine 200 once daily | 1.02 (0.97, 1.08) | 1.01 (0.98, 1.04) | 1.06 (1.01, 1.11) | |||||
| tenofovir DF 300 once daily | 1.36 (1.25, 1.47) | 1.35 (1.29, 1.42) | 1.45 (1.39, 1.51) | |||||
| Emtricitabine/ rilpivirine/ tenofovir DF♠ | emtricitabine 200 once daily | 400 once daily | 100 once daily | 24 | 0.95 (0.90, 1.00) | 0.99 (0.97, 1.02) | 1.05 (0.99, 1.11) | |
| rilpivirine 25 once daily | 0.93 (0.88, 0.98) | 0.95 (0.90, 1.00) | 0.96 (0.90, 1.03) | |||||
| tenofovir DF 300 once daily | 1.44 (1.33, 1.55) | 1.40 (1.34, 1.46) | 1.84 (1.76, 1.92) | |||||
| Norelgestromin | norgestimate 0.180/0.215/0.25/ethinyl estradiol 0.025 once daily | ND | 100 once daily | 13 | 0.97 (0.88, 1.07) | 0.90 (0.82, 0.98) | 0.92 (0.83, 1.03) | |
| 400 once daily | ND | 15 | 1.07 (0.94, 1.22) | 1.06 (0.92, 1.21) | 1.07 (0.89, 1.28) | |||
| Norgestrel | ND | 100 once daily | 13 | 0.96 (0.78, 1.19) | 0.91 (0.73, 1.15) | 0.92 (0.73, 1.18) | ||
| 400 once daily | ND | 15 | 1.18 (0.99, 1.41) | 1.19 (0.98, 1.45) | 1.23 (1.00, 1.51) | |||
| Ethinyl estradiol | ND | 100 once daily | 12 | 1.39 (1.17, 1.66) | 1.04 (0.87, 1.24) | 0.83 (0.65, 1.06) | ||
| 400 once daily | ND | 15 | 1.15 (0.97, 1.36) | 1.09 (0.94, 1.26) | 0.99 (0.80, 1.23) | |||
| Pravastatin | 40 single dose | ND | 100 once daily | 18 | 1.28 (1.08, 1.52) | 1.35 (1.18, 1.54) | NA | |
| Rosuvastatin | 10 single dose | ND | 100 once daily | 18 | 2.61 (2.32, 2.92) | 2.69 (2.46, 2.94) | NA | |
| Raltegravir + emtricitabine/ tenofovir DF | emtricitabine 200 once daily | 400 once daily | 100 once daily | 30 | 1.08 (1.04, 1.12) | 1.05 (1.03, 1.07) | 1.02 (0.97, 1.08) | |
| tenofovir DF 300 once daily | 1.46 (1.39, 1.54) | 1.40 (1.34, 1.45) | 1.70 (1.61, 1.79) | |||||
| raltegravir 400 twice daily | 1.03 (0.74, 1.43) | 0.97 (0.73, 1.28) | 0.79 (0.42, 1.48) | |||||
| Tacrolimus | 5 single dose | 400 single dose | ND | 16 | 0.73 (0.59, 0.90) | 1.09 (0.84, 1.40) | NA | |
No effect on the pharmacokinetic parameters of the following coadministered drugs was observed or is expected with EPCLUSA (buprenorphine/naloxone, dolutegravir, lopinavir/ritonavir, or methadone) or its components sofosbuvir (cyclosporine) or velpatasvir (cyclosporine).
12.4 Microbiology
Mechanism of Action
Sofosbuvir is an inhibitor of the HCV NS5B RNA-dependent RNA polymerase, which is required for viral replication. Sofosbuvir is a nucleotide prodrug that undergoes intracellular metabolism to form the pharmacologically active uridine analog triphosphate (GS-461203), which can be incorporated into HCV RNA by the NS5B polymerase and acts as a chain terminator. In a biochemical assay, GS-461203 inhibited the polymerase activity of the recombinant NS5B from HCV genotype 1b, 2a, 3a, and 4a with an IC50 value ranging from 0.36 to 3.3 micromolar. GS-461203 is neither an inhibitor of human DNA and RNA polymerases nor an inhibitor of mitochondrial RNA polymerase.
Velpatasvir is an inhibitor of the HCV NS5A protein, which is required for viral replication. Resistance selection in cell culture and cross-resistance studies indicate velpatasvir targets NS5A as its mode of action.
Antiviral Activity
The EC50 values of sofosbuvir and velpatasvir against full-length or chimeric replicons encoding NS5B and NS5A sequences from the laboratory strains are presented in Table 10. The EC50 values of sofosbuvir and velpatasvir against clinical isolates are presented in Table 11.
| Replicon Genotype | Sofosbuvir EC50, nM* | Velpatasvir EC50, nM* |
|---|---|---|
| NA = not available. | ||
| ||
| 1a | 40 | 0.014 |
| 1b | 110 | 0.016 |
| 2a | 50 | 0.005–0.016† |
| 2b | 15‡ | 0.002–0.006† |
| 3a | 50 | 0.004 |
| 4a | 40 | 0.009 |
| 4d | 33.4 | 0.004 |
| 5a | 15‡ | 0.021–0.054§ |
| 6a | 14–25‡ | 0.006–0.009 |
| 6e | NA | 0.130§ |
| Replicon Genotype | Replicons Containing NS5B from Clinical Isolates | Replicons Containing NS5A from Clinical Isolates | ||
|---|---|---|---|---|
| Number of clinical isolates | Median sofosbuvir EC50, nM (range) | Number of clinical isolates | Median velpatasvir EC50, nM (range) | |
| NA = not available. | ||||
| 1a | 67 | 62 (29–128) | 23 | 0.019 (0.011–0.078) |
| 1b | 29 | 102 (45–170) | 34 | 0.012 (0.005–0.500) |
| 2a | 1 | 28 | 8 | 0.011 (0.006–0.364) |
| 2b | 14 | 30 (14–81) | 16 | 0.002 (0.0003–0.007) |
| 3a | 106 | 81 (24–181) | 38 | 0.005 (0.002–1.871) |
| 4a | NA | NA | 5 | 0.002 (0.001–0.004) |
| 4d | NA | NA | 10 | 0.007 (0.004–0.011) |
| 4r | NA | NA | 7 | 0.003 (0.002–0.006) |
| 5a | NA | NA | 42 | 0.005 (0.001–0.019) |
| 6a | NA | NA | 26 | 0.007 (0.0005–0.113) |
| 6e | NA | NA | 15 | 0.024 (0.005–0.433) |
Velpatasvir was not antagonistic in reducing HCV RNA levels in replicon cells when combined with sofosbuvir or interferon-α, ribavirin, an HCV NS3/4A protease inhibitor, or HCV NS5B non-nucleoside inhibitors.
Resistance
In Cell Culture
HCV replicons with reduced susceptibility to sofosbuvir have been selected in cell culture for multiple genotypes including 1b, 2a, 2b, 3a, 4a, 5a, and 6a. Reduced susceptibility to sofosbuvir was associated with the NS5B substitution S282T in all replicon genotypes examined. An M289L substitution developed along with the S282T substitution in genotype 2a, 5, and 6 replicons. Site-directed mutagenesis of the S282T substitution in replicons of genotypes 1 to 6 conferred 2- to 18-fold reduced susceptibility to sofosbuvir.
HCV genotype 1a, 1b, 2a, 3a, 4a, 5a, and 6a replicon variants with reduced susceptibility to velpatasvir were selected in cell culture. Variants developed amino acid substitutions at NS5A resistance-associated positions 24, 28, 30, 31, 32, 58, 92, and 93. Phenotypic analysis of site-directed mutant replicons of the selected NS5A substitutions showed that single and double combinations of L31V and Y93H/N in genotype 1a, the combination of L31V + Y93H in genotype 1b, Y93H/S in genotype 3a, and L31V and P32A/L/Q/R in genotype 6 conferred greater than 100-fold reduction in velpatasvir susceptibility. In the genotype 2a replicon, the single mutants F28S and Y93H showed 91-fold and 46-fold reduced susceptibility to velpatasvir, respectively. The single mutant Y93H conferred 3-fold reduced susceptibility to velpatasvir in genotype 4a replicons. Combinations of these NS5A substitutions often showed greater reductions in susceptibility to velpatasvir than single substitutions alone.
In Clinical Trials
Studies in Subjects without Cirrhosis and Subjects with Compensated Cirrhosis
In a pooled analysis of subjects without cirrhosis or with compensated cirrhosis who received EPCLUSA for 12 weeks in Phase 3 trials (ASTRAL-1, ASTRAL-2, and ASTRAL-3), 12 subjects (2 with genotype 1 [1a, 1c/h] and 10 with genotype 3a) qualified for resistance analysis due to virologic failure. No subjects with genotype 2, 4, 5, or 6 HCV infection experienced virologic failure.
Of the 2 genotype 1 virologic failure subjects, 1 subject had virus with emergent NS5A resistance substitution Y93N and the other had virus with emergent NS5A resistance substitutions Y93H and low-level K24M/T and L31I/V at virologic failure. The latter subject had genotype 1c/h virus at baseline harboring NS5A resistance polymorphisms (Q30R, L31M, H58P) relative to genotype 1a. No sofosbuvir NS5B nucleoside analog resistance-associated substitutions were observed at failure in the 2 subjects.
Of the 10 genotype 3a virologic failure subjects, NS5A resistance substitution Y93H was observed in all 10 subjects at failure (7 subjects had Y93H emerge post-treatment and 3 subjects had Y93H at baseline and post-treatment). Treatment-emergent sofosbuvir NS5B substitutions L314F (n=2) and L314I (n=1) were observed at high frequency (greater than or equal to 15%) in the NS5B polymerase in 3 genotype 3a subjects who relapsed: one in the EPCLUSA group and two in the sofosbuvir plus ribavirin 24-week group. In addition, low frequency (less than 4%) treatment-emergent L314P was detected in 2 genotype 3a subjects who relapsed, including one subject in the sofosbuvir plus ribavirin 24-week group in ASTRAL-3 and one in the EPCLUSA group in ASTRAL-4. The clinical significance of this substitution is unknown.
In Trial 2104 (liver transplant recipients), there were 2 virologic failures (1 subject with genotype 1a and 1 subject with genotype 3b). The genotype 1a virologic failure subject had virus with an NS5A K24R polymorphism at baseline and relapse, and treatment-emergent NS5A L31V. The genotype 3b virologic failure subject had virus at baseline and relapse with NS5A polymorphisms A30K+L31M, which are predominant in this subtype; treatment-emergent NS5B S282T was detected at relapse.
Studies in Subjects with Decompensated Cirrhosis
In the ASTRAL-4 trial in subjects with decompensated cirrhosis who received EPCLUSA with ribavirin for 12 weeks, 3 subjects (1 with genotype 1a and 2 with genotype 3a) qualified for resistance analysis due to virologic failure. No subjects with genotype 2 or 4 HCV infection who received EPCLUSA with ribavirin for 12 weeks experienced virologic failure.
The genotype 1 virologic failure subject had no NS5A or NS5B resistance substitutions at failure.
The 2 genotype 3a virologic failure subjects had the NS5A resistance substitutions Y93H and either low-level M28V or S38P emerge at failure. One of these subjects also developed low levels (less than 5%) of NS5B nucleoside analog inhibitor resistance substitutions N142T and E237G at failure.
In the ASTRAL-4 trial, 2 subjects treated with EPCLUSA for 12 or 24 weeks without ribavirin had emergent sofosbuvir NS5B resistance-associated substitutions S282T at low levels (less than 5%) along with L159F. EPCLUSA for 12 or 24 weeks without ribavirin is not recommended in patients with decompensated cirrhosis.
Persistence of Resistance-Associated Substitutions
No data are available on the persistence of sofosbuvir or velpatasvir resistance-associated substitutions. NS5A resistance-associated substitutions observed with administration of other NS5A inhibitors have been found to persist for longer than 1 year in most patients. The long-term clinical impact of the emergence or persistence of virus containing sofosbuvir or velpatasvir resistance-associated substitutions is unknown.
Effect of Baseline HCV Polymorphisms on Treatment Response
Adults
Analyses were conducted to explore the association between relapse rates and pre-existing baseline NS5A resistance-associated polymorphisms (RAPs) (any change from reference at NS5A amino acid positions 24, 28, 30, 31, 58, 92, or 93) identified by population or deep sequencing analysis at a sensitivity threshold of 15% or higher for subjects without cirrhosis or with compensated cirrhosis in ASTRAL-1, ASTRAL-2, and ASTRAL-3 and subjects with decompensated cirrhosis in ASTRAL-4.
Studies in Subjects without Cirrhosis and Subjects with Compensated Cirrhosis
Among the subjects who received treatment with EPCLUSA for 12 weeks, 18% (37/209), 32% (38/117), 64% (149/232), 20% (56/274), 63% (73/115), 9% (3/34), and 83% (35/42) of subjects with genotype 1a, 1b, 2, 3, 4, 5, and 6 HCV, respectively, had baseline virus with NS5A RAPs.
Genotype 1: Among the 75 genotype 1 subjects who had baseline NS5A RAPs, one subject (1%) with Q30R, L31M, and H58P polymorphisms at baseline and compensated cirrhosis relapsed.
Genotype 3: Among the 56 genotype 3 subjects in ASTRAL-3 who had baseline NS5A RAPs, 4 subjects (7%) relapsed (3 with baseline Y93H and 1 with baseline A30K). Overall, 20% (3/15) of genotype 3 subjects with the Y93H polymorphism at baseline relapsed.
In a pooled analysis of clinical trials in subjects with HCV genotype 3 infection, the prevalence of the baseline NS5A Y93H polymorphism was 6% (104/1842). Among HCV genotype 3 infected subjects with the Y93H polymorphism who were treated with EPCLUSA for 12 weeks, 7% (2/28) of subjects without cirrhosis relapsed, and 40% (6/15) of subjects with compensated cirrhosis experienced virologic failure (5 relapse, 1 on-treatment).
Genotypes 2, 4, 5, and 6: The presence of baseline NS5A RAPs did not affect relapse rates for subjects with genotypes 2, 4, 5, and 6, because all achieved sustained virologic response (SVR) 12.
SVR12 was achieved in all 77 subjects who had baseline NS5B nucleoside analog inhibitor resistance polymorphisms including N142T, L159F, E/N237G, C/M289L/I, L320F/I/V, V321A/I, and S282G + V321I. The sofosbuvir NS5B nucleoside analog inhibitor resistance substitution S282T was not detected in the baseline NS5B sequence of any subject using 1% deep sequencing cutoff in Phase 3 trials.
Studies in Subjects with Decompensated Cirrhosis
In ASTRAL-4, the prevalence of NS5A RAPs at baseline was 24% (48/198), 60% (6/10), 11% (4/37), and 63% (5/8) in GT1, GT2, GT3, and GT4 HCV subjects, respectively. No subjects with genotypes 2, 4 and 6 relapsed. There were no subjects with genotype 5 in this trial.
For genotype 1 subjects, the overall relapse rates were numerically lower for the 12-week EPCLUSA with ribavirin group (2%; 1/66) compared to EPCLUSA 12-week and 24-week treatment groups. For subjects with NS5A RAPs, relapse rates were 0% (0/17) compared to 2% (1/49) for subjects without NS5A RAPs in the 12-week EPCLUSA with ribavirin containing group.
For genotype 3 subjects, overall virologic failure rates were numerically lower for the 12-week EPCLUSA with ribavirin group (15%; 2/13) compared to EPCLUSA 12-week and 24-week treatment groups. There are insufficient data to determine the impact of HCV NS5A RAPs in genotype 3 subjects with decompensated cirrhosis.
Three subjects in the EPCLUSA with ribavirin 12-week group had baseline NS5B nucleoside analog inhibitor polymorphisms (N142T and L159F) using 1% deep sequencing cutoff and all 3 subjects achieved SVR12.
Cross Resistance
Both sofosbuvir and velpatasvir were fully active against substitutions associated with resistance to other classes of direct-acting antivirals with different mechanisms of action, such as NS5B non-nucleoside inhibitors and NS3 protease inhibitors. The efficacy of EPCLUSA has not been established in patients who have previously failed treatment with other regimens that include an NS5A inhibitor.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
Sofosbuvir: Sofosbuvir was not genotoxic in a battery of in vitro or in vivo assays, including bacterial mutagenicity, chromosome aberration using human peripheral blood lymphocytes, and in vivo mouse micronucleus assays.
Sofosbuvir was not carcinogenic in a 2-year mouse study (up to 200 mg/kg/day in males and 600 mg/kg/day in females) and in a 2-year rat study (up to 750 mg/kg/day), resulting in exposures of the predominant circulating metabolite GS-331007 of approximately 3 and 15 times (in mice) and 7 and 9 times (in rats), in males and females, respectively, the exposure in humans at the recommended human dose (RHD).
Velpatasvir: Velpatasvir was not genotoxic in a battery of in vitro or in vivo assays, including bacterial mutagenicity, chromosome aberration using human peripheral blood lymphocytes, and in vivo rat micronucleus assays.
Velpatasvir was not carcinogenic in a 6-month rasH2 transgenic mouse study (up to 1000 mg/kg/day) and a 2-year rat carcinogenicity study (up to 200 mg/kg/day). The exposure of VEL in the 2-year rat study was approximately 6 times the exposure in humans at the RHD.
14. Clinical Studies
14.1 Description of Clinical Trials
Table 12 presents the clinical trial design including different treatment groups that were conducted with EPCLUSA with and without ribavirin in subjects with chronic hepatitis C (HCV) genotype 1, 2, 3, 4, 5, and 6 infection. For detailed description of trial design and recommended regimen and duration [see Dosage and Administration (2.2, 2.3, and 2.4) and Clinical Studies (14.2, 14.3, 14.4, 14.5, 14.6, 14.7, and 14.8)].
| Trial | Population | EPCLUSA and Comparator Groups (Number of Subjects Treated) |
|---|---|---|
| TN = treatment-naïve subjects; SOF = sofosbuvir; RBV = ribavirin; CP = Child-Pugh; ESRD = End Stage Renal Disease; PWID = People Who Inject Drugs; MAT = Medication-Assisted Treatment. | ||
| ||
| ASTRAL-1*
(NCT02201940) | Genotype 1, 2, 4, 5, and 6 TN and TE†, without cirrhosis or with compensated cirrhosis | EPCLUSA 12 weeks (624) Placebo 12 weeks (116) |
| ASTRAL-2‡
(NCT02220998) | Genotype 2 TN and TE†, without cirrhosis or with compensated cirrhosis | EPCLUSA 12 weeks (134) SOF + RBV 12 weeks (132) |
| ASTRAL-3‡
(NCT02201953) | Genotype 3 TN and TE†, without cirrhosis or with compensated cirrhosis | EPCLUSA 12 weeks (277) SOF + RBV 24 weeks (275) |
| ASTRAL-5‡
(NCT02480712) | Genotype 1, 2, 3, 4, 5, and 6 HCV/HIV-1 coinfected TN and TE†, without cirrhosis or with compensated cirrhosis | EPCLUSA 12 weeks (106) |
| ASTRAL-4‡
(NCT02201901) | Genotype 1, 2, 3, 4, 5, and 6 TN and TE†, with CP class B decompensated cirrhosis | EPCLUSA 12 weeks (90) EPCLUSA + RBV 12 weeks (87) EPCLUSA 24 weeks (90) |
| 2104‡
(NCT02781571) | Genotype 1, 2, 3, and 4 TN and TE§ liver transplant recipients, without cirrhosis or with compensated cirrhosis | EPCLUSA 12 weeks (79) |
| 4062‡
(NCT03036852) | Genotype 1, 2, 3, 4, and 6 TN and TE¶ without cirrhosis or with compensated cirrhosis, with ESRD requiring dialysis | EPCLUSA 12 weeks (59) |
| SIMPLIFY‡
(NCT02336139) | Genotype 1, 2, 3, and 4 PWID, including those on MAT for opioid use disorder, without cirrhosis or with compensated cirrhosis | EPCLUSA 12 weeks (103) |
| 1143‡
(NCT03022981) | Genotype 1, 2, 3, 4, and 6 TN and TE# pediatric subjects 3 years of age and older | EPCLUSA 12 weeks (214) |
The ribavirin dosage was weight-based (1000 mg daily administered in two divided doses for subjects less than 75 kg and 1200 mg for those greater than or equal to 75 kg) and administered in two divided doses when used in combination with sofosbuvir in the ASTRAL-2 and ASTRAL-3 trials or in combination with EPCLUSA in the ASTRAL-4 trial. Ribavirin dosage adjustments were performed according to the ribavirin prescribing information. Serum HCV RNA values were measured during the clinical trials using the COBAS AmpliPrep/COBAS Taqman HCV test (version 2.0) with a lower limit of quantification (LLOQ) of 15 IU/mL. SVR12, defined as HCV RNA less than LLOQ at 12 weeks after the cessation of treatment, was the primary endpoint in all the trials. Relapse is defined as HCV RNA greater than or equal to LLOQ during the post-treatment period after having achieved HCV RNA less than LLOQ at the end of treatment. On-treatment virologic failure is defined as breakthrough, rebound, or non-response.
14.2 Clinical Trials in Subjects without Cirrhosis and Subjects with Compensated Cirrhosis
Genotype 1, 2, 4, 5, and 6 HCV Infected Adults (ASTRAL-1)
ASTRAL-1 was a randomized, double-blind, placebo-controlled trial that evaluated 12 weeks of treatment with EPCLUSA compared with 12 weeks of placebo in subjects with genotype 1, 2, 4, 5, or 6 HCV infection without cirrhosis or with compensated cirrhosis. Subjects with genotype 1, 2, 4, or 6 HCV infection were randomized in a 5:1 ratio to treatment with EPCLUSA or placebo for 12 weeks. Subjects with genotype 5 HCV infection were enrolled to the EPCLUSA group. Randomization was stratified by HCV genotype (1, 2, 4, 6, and indeterminate) and the presence or absence of compensated cirrhosis.
Demographics and baseline characteristics were balanced between the EPCLUSA and placebo group. Of the 740 treated subjects, the median age was 56 years (range: 18 to 82); 60% of the subjects were male; 79% were White, 9% were Black; 21% had a baseline body mass index at least 30 kg/m2; the proportions of subjects with genotype 1, 2, 4, 5, or 6 HCV infection were 53%, 17%, 19%, 5%, and 7%, respectively; 69% had non-CC IL28B alleles (CT or TT); 74% had baseline HCV RNA levels at least 800,000 IU/mL; 19% had compensated cirrhosis; and 32% were treatment-experienced.
Table 13 presents SVR12 and other virologic outcomes in EPCLUSA-treated subjects in the ASTRAL-1 trial by HCV genotype. No subjects in the placebo group achieved SVR12.
| EPCLUSA 12 Weeks (N=624) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Total (all GTs) (N=624) | GT-1 | GT-2 (N=104) | GT-4 (N=116) | GT-5 (N=35) | GT-6 (N=41) | |||||
| GT-1a (N=210) | GT-1b (N=118) | Total (N=328) | ||||||||
| GT = genotype; no subjects in the placebo group achieved SVR12. | ||||||||||
| ||||||||||
| SVR12 | 99% (618/624) | 98% (206/210) | 99% (117/118) | 98% (323/328) | 100% (104/104) | 100% (116/116) | 97% (34/35) | 100% (41/41) | ||
| Outcome for Subjects without SVR | ||||||||||
| On-Treatment Virologic Failure | 0/624 | 0/210 | 0/118 | 0/328 | 0/104 | 0/116 | 0/35 | 0/41 | ||
| Relapse* | <1% (2/623) | <1% (1/209) | 1% (1/118) | 1% (2/327) | 0/104 | 0/116 | 0/35 | 0/41 | ||
| Other† | 1% (4/624) | 1% (3/210) | 0/118 | 1% (3/328) | 0/104 | 0/116 | 3% (1/35) | 0/41 | ||
Genotype 2 HCV Infected Adults (ASTRAL-2)
ASTRAL-2 was a randomized, open-label trial that evaluated 12 weeks of treatment with EPCLUSA compared with 12 weeks of treatment with SOF with ribavirin in subjects with genotype 2 HCV infection. Subjects were randomized in a 1:1 ratio to the treatment groups. Randomization was stratified by the presence or absence of compensated cirrhosis and prior treatment experience (treatment-naïve vs treatment-experienced).
Demographics and baseline characteristics were balanced across the two treatment groups. Of the 266 treated subjects, the median age was 58 years (range: 23 to 81); 59% of the subjects were male; 88% were White; 7% were Black; 33% had a baseline body mass index at least 30 kg/m2; 62% had non-CC IL28B alleles (CT or TT); 80% had baseline HCV RNA levels at least 800,000 IU/mL; 14% had compensated cirrhosis; and 15% were treatment-experienced.
Table 14 presents SVR12 and other virologic outcomes from the ASTRAL-2 trial.
| EPCLUSA 12 Weeks (N=134) | SOF + RBV 12 Weeks (N=132) | |
|---|---|---|
| SOF = sofosbuvir; RBV = ribavirin. | ||
| ||
| SVR12 | 99% (133/134) | 94% (124/132) |
| Treatment difference +5.2%; 95% confidence interval (+0.2% to +10.3%) | ||
| Outcome for subjects without SVR | ||
| On-Treatment Virologic Failure | 0/134 | 0/132 |
| Relapse* | 0/133 | 5% (6/132) |
| Other† | 1% (1/134) | 2% (2/132) |
Genotype 3 HCV Infected Adults (ASTRAL-3)
ASTRAL-3 was a randomized, open-label trial that evaluated 12 weeks of treatment with EPCLUSA compared with 24 weeks of treatment with SOF with ribavirin in subjects with genotype 3 HCV infection. Subjects were randomized in a 1:1 ratio to the treatment groups. Randomization was stratified by the presence or absence of compensated cirrhosis and prior treatment experience (treatment-naïve vs treatment-experienced).
Demographics and baseline characteristics were balanced across the treatment groups. Of the 552 treated subjects, the median age was 52 years (range: 19 to 76); 62% of the subjects were male; 89% were White; 9% were Asian; 20% had a baseline body mass index at least 30 kg/m2; 61% had non-CC IL28B alleles (CT or TT); 70% had baseline HCV RNA levels at least 800,000 IU/mL; 30% had compensated cirrhosis; and 26% were treatment-experienced.
Table 15 presents SVR12 and other virologic outcomes from the ASTRAL-3 trial.
| EPCLUSA 12 Weeks (N=277) | SOF + RBV 24 Weeks (N=275) | |
|---|---|---|
| SOF = sofosbuvir; RBV = ribavirin. | ||
| ||
| SVR12 | 95% (264/277) | 80% (221/275) |
| Treatment difference +14.8%; 95% confidence interval (+9.6% to +20.0%) | ||
| Outcome for subjects without SVR | ||
| On-Treatment Virologic Failure | 0/277 | <1% (1/275) |
| Relapse* | 4% (11/276) | 14% (38/272) |
| Other† | 1% (2/277) | 5% (15/275) |
SVR12 for selected subgroups are presented in Table 16.
| EPCLUSA 12 Weeks | SOF + RBV 24 Weeks* | |||||
|---|---|---|---|---|---|---|
| Treatment-Naïve (N=206) | Treatment-Experienced (N=71) | Treatment-Naïve (N=201) | Treatment-Experienced (N=69) | |||
| SOF = sofosbuvir; RBV = ribavirin. | ||||||
| ||||||
| Without cirrhosis | 98% (160/163) | 94% (31/33)† | 90% (141/156) | 71% (22/31) | ||
| With compensated cirrhosis | 93% (40/43) | 89% (33/37) | 73% (33/45) | 58% (22/38) | ||
14.3 Clinical Trial in Subjects Coinfected with HCV and HIV-1
ASTRAL-5 was an open-label trial that evaluated 12 weeks of treatment with EPCLUSA in subjects with genotype 1, 2, 3, 4, 5, or 6 HCV infection who were coinfected with HIV-1. Subjects were on a stable HIV-1 antiretroviral therapy that included emtricitabine/tenofovir disoproxil fumarate or abacavir/lamivudine administered with atazanavir/ritonavir, darunavir/ritonavir, lopinavir/ritonavir, rilpivirine, raltegravir or elvitegravir/cobicistat.
Of the 106 treated subjects, the median age was 57 years (range: 25 to 72); 86% of the subjects were male; 51% were White; 45% were Black; 22% had a baseline body mass index at least 30 kg/m2; the proportions of patients with genotype 1, 2, 3, or 4 HCV infection were 74%, 10%, 11%, and 5%, respectively; no subjects with genotype 5 or 6 HCV were treated with EPCLUSA; 77% had non-CC IL28B alleles (CT or TT); 74% had baseline HCV RNA levels of at least 800,000 IU/mL; 18% had compensated cirrhosis; and 29% were treatment experienced. The overall mean CD4+ count was 598 cells/µL (range: 183−1513 cells/µL) and 57% of subjects had CD4+ counts > 500 cells/μL.
Table 17 presents the SVR12 for the ASTRAL-5 trial by HCV genotype.
| EPCLUSA 12 Weeks (N=106) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Total (all GTs) (N=106) | GT-1 | GT-2 (N=11) | GT-3 (N=12) | GT-4 (N=5) | |||||
| GT-1a (N=66) | GT-1b (N=12) | Total (N=78) | |||||||
| SVR12 | 95% (101/106) | 95% (63/66) | 92% (11/12) | 95% (74/78) | 100% (11/11) | 92% (11/12) | 100% (5/5) | ||
| Outcome for Subjects without SVR | |||||||||
| On-Treatment Virologic Failure | 0/106 | 0/66 | 0/12 | 0/78 | 0/11 | 0/12 | 0/5 | ||
| Relapse* | 2% (2/103) | 3% (2/65) | 0/11 | 3% (2/76) | 0/11 | 0/11 | 0/5 | ||
| Other† | 3% (3/106) | 2% (1/66) | 8% (1/12) | 3% (2/78) | 0/11 | 8% (1/12) | (0/5) | ||
| |||||||||
No subject had HIV-1 rebound during treatment and CD4+ counts were stable during treatment.
14.4 Clinical Trials in Subjects with Decompensated Cirrhosis
ASTRAL-4 was a randomized, open-label trial in subjects with genotype 1, 2, 3, 4, 5, or 6 HCV infection and Child-Pugh B cirrhosis at screening. Subjects were randomized in a 1:1:1 ratio to treatment with EPCLUSA for 12 weeks (N=90), EPCLUSA with ribavirin for 12 weeks (N=87), or EPCLUSA for 24 weeks (N=90). Randomization was stratified by HCV genotype (1, 2, 3, 4, 5, 6, and indeterminate).
Demographics and baseline characteristics were balanced across the treatment groups. Of the 267 treated subjects, the median age was 59 years (range: 40 to 73); 70% of the subjects were male; 90% were White; 6% were Black; 42% had a baseline body mass index at least 30 kg/m2. The proportions of subjects with genotype 1, 2, 3, 4, or 6 HCV were 78%, 4%, 15%, 3%, and less than 1% (1 subject), respectively. No subjects with genotype 5 HCV infection were enrolled. 76% had non-CC IL28B alleles (CT or TT); 56% had baseline HCV RNA levels at least 800,000 IU/mL; 55% were treatment-experienced; and 95% of subjects had Model for End Stage Liver Disease (MELD) score less than or equal to 15 at baseline. Although all subjects had Child-Pugh B cirrhosis at screening, 6% and 4% of subjects were assessed to have Child-Pugh A and Child-Pugh C cirrhosis, respectively, on the first day of treatment.
Treatment with EPCLUSA with ribavirin for 12 weeks resulted in numerically higher SVR12 rates than treatment with EPCLUSA for 12 weeks or 24 weeks. Because EPCLUSA with ribavirin for 12 weeks is the recommended dosage regimen, the results of the 12- and 24-week EPCLUSA treatment groups are not presented.
Table 18 presents the SVR12 for subjects treated with EPCLUSA with ribavirin for 12 weeks in the ASTRAL-4 trial by HCV genotype. No subjects with genotype 5 or 6 HCV were treated with EPCLUSA with ribavirin for 12 weeks.
| EPCLUSA + RBV 12 Weeks (N=87) | |||
|---|---|---|---|
| SVR12 | Virologic Failure (relapse and on-treatment failure) | ||
| RBV = ribavirin. | |||
| |||
| Overall SVR12* | 94% (82/87) | 3% (3/87) | |
| Genotype 1 | 96% (65/68) | 1% (1/68)† | |
| Genotype 1a | 94% (51/54) | 2% (1/54)† | |
| Genotype 1b | 100% (14/14) | 0% (0/14) | |
| Genotype 3 | 85% (11/13) | 15% (2/13)‡ | |
All subjects with genotype 2 (N=4) and genotype 4 (N=2) HCV infection treated with EPCLUSA and ribavirin achieved SVR12.
14.5 Clinical Trial in Adult Liver Transplant Recipients without Cirrhosis and with Compensated Cirrhosis
Trial 2104 was an open-label clinical trial that evaluated 12 weeks of treatment with EPCLUSA in 79 HCV-infected treatment-naïve and previously treated adult subjects who had undergone liver transplantation. The proportions of subjects with genotype 1, 2, 3, or 4 HCV infection were 47%, 4%, 44%, and 5%, respectively. The median age was 62 years (range: 45 to 81); 81% were male; 82% were White; 3% were Black; and 15% were Asian; 28% had a baseline body mass index at least 30 kg/m2. At baseline, 18% had compensated cirrhosis, and 60% were treatment experienced (subjects with prior exposure to any HCV NS5A inhibitor were excluded). Immunosuppressants allowed for coadministration were tacrolimus, mycophenolate mofetil, cyclosporine, and azathioprine. The overall SVR12 rate was 96% (76/79). Of the subjects completing 12 weeks of EPCLUSA, 2 subjects experienced virologic relapse.
14.6 Clinical Trial in Subjects with Severe Renal Impairment Requiring Dialysis
Trial 4062 was an open-label clinical trial that evaluated 12 weeks of treatment with EPCLUSA in 59 HCV-infected adults with ESRD requiring dialysis. The proportions of subjects with genotype 1, 2, 3, 4, 6 or indeterminate HCV infection were 42%,12%, 27%, 7%, 3%, and 8%, respectively. At baseline, 29% of subjects had cirrhosis, 22% were treatment-experienced (subjects with prior exposure to any HCV NS5A inhibitor were excluded), 92% were on hemodialysis, and 8% were on peritoneal dialysis; mean duration on dialysis was 7 years (range: 0 to 40 years). The overall SVR rate was 95% (56/59). Of the subjects completing 12 weeks of EPCLUSA, 1 subject experienced virologic relapse.
14.7 Clinical Trial in People who Inject Drugs (PWID), Including Those on Medication-Assisted Treatment (MAT) for Opioid Use Disorder
SIMPLIFY was an open-label Phase 2 clinical trial that evaluated 12 weeks of treatment with EPCLUSA in 103 HCV-infected PWID (defined as self-reported injection drug use within previous 6 months), including 58 subjects on MAT for opioid use disorder. The proportions of subjects with genotype 1, 2, 3, and 4 HCV infection were 35%, 5%, 58%, and 2%, respectively. The median age was 48 years (range: 24 to 67); 71% were male; 89% were White; and 2% were Black. At baseline, 74% and 26% of subjects reported injection drug use or daily injection drug use, respectively, in the past month; 56% had baseline HCV RNA levels at least 800,000 IU/mL; 10% had compensated cirrhosis; and all subjects were naïve to prior exposure with SOF or an HCV NS5A inhibitor. Subjects on MAT for opioid use disorder reported concomitant use of methadone (76%) and buprenorphine naloxone (17%) with EPCLUSA. The overall SVR rate was 94% (97/103). One subject completed EPCLUSA treatment and was re-infected with a phylogenetically different virus; the other 5 subjects who did not achieve SVR12 did not meet virologic failure criteria.
14.8 Clinical Trial in Pediatric Subjects
The efficacy of EPCLUSA once daily for 12 weeks was evaluated in an open-label trial (Study 1143) in 214 genotype 1, 2, 3, 4, or 6 HCV treatment-naïve (N=188) or treatment-experienced (N=26) pediatric subjects 3 years of age and older without cirrhosis or with compensated cirrhosis.
Subjects 12 Years to <18 Years of Age: EPCLUSA was evaluated in 102 subjects 12 years to <18 years of age with genotype 1, 2, 3, 4, or 6 HCV infection. Among these subjects, 80 (78%) were treatment-naïve and 22 (22%) were treatment-experienced. The median age was 15 years (range: 12 to 17); 51% of the subjects were female; 73% were White, 9% were Black, and 11% were Asian; 14% were Hispanic/Latino; mean body mass index was 23 kg/m2 (range: 13 to 49 kg/m2); mean weight was 61 kg (range: 22 to 147 kg); 58% had baseline HCV RNA levels greater than or equal to 800,000 IU/mL; the proportions of subjects with genotype 1, 2, 3, 4, or 6 HCV infection were 74%, 6%, 12%, 2%, and 6%, respectively; no subjects had known cirrhosis. The majority of subjects (89%) had been infected through vertical transmission.
The SVR rate was 93% (71/76) in subjects with genotype 1 HCV infection and 100% in subjects with genotype 2 (6/6), genotype 3 (12/12), genotype 4 (2/2), and genotype 6 (6/6) HCV infection. One subject discontinued treatment at Week 4 and subsequently relapsed; the other four subjects who did not achieve SVR12 did not meet virologic failure criteria (lost to follow-up).
Subjects 6 Years to <12 Years of Age: EPCLUSA was evaluated in 71 subjects 6 years to <12 years of age with genotype 1, 2, 3, or 4 HCV infection. Among these subjects, 67 (94%) were treatment-naïve and 4 (6%) were treatment-experienced. The median age was 8 years (range: 6 to 11); 54% of the subjects were female; 90% were White, 6% were Black, and 1% were Asian; 10% were Hispanic/Latino; mean body mass index was 17 kg/m2 (range: 13 to 31 kg/m2); mean weight was 30 kg (range: 18 to 78 kg); 48% had baseline HCV RNA levels greater than or equal to 800,000 IU per mL; the proportions of subjects with genotype 1, 2, 3, or 4 HCV infection were 76%, 3%, 15%, and 6%, respectively; no subjects had known cirrhosis. The majority of subjects (94%) had been infected through vertical transmission.
The SVR rate was 93% (50/54) in subjects with genotype 1 HCV infection, 91% (10/11) in subjects with genotype 3 HCV infection, and 100% in subjects with genotype 2 (2/2) and genotype 4 (4/4) HCV infection. One subject had on-treatment virologic failure; the other four subjects who did not achieve SVR12 did not meet virologic failure criteria (lost to follow-up).
Subjects 3 Years to <6 Years of Age: EPCLUSA was evaluated in 41 treatment-naïve subjects 3 years to <6 years of age with genotype 1, 2, 3, or 4 HCV infection. The median age was 4 years (range: 3 to 5); 59% of the subjects were female; 78% were White, 7% were Black; 10% were Hispanic/Latino; mean body mass index was 17.0 kg/m2 (range: 13.9 to 22.0 kg/m2); mean weight was 19 kg (range: 13 to 35 kg); 49% had baseline HCV RNA levels greater than or equal to 800,000 IU per mL; the proportions of subjects with genotype 1, 2, 3, or 4 HCV infection were 78%, 15%, 5%, and 2%, respectively; no subjects had known cirrhosis. The majority of subjects (98%) had been infected through vertical transmission.
The SVR12 rate among all subjects was 83% (34/41); with 88% (28/32) in subjects with genotype 1 HCV infection, 50% (3/6) in subjects with genotype 2 HCV infection, and 100% in subjects with genotype 3 (2/2) and genotype 4 (1/1) HCV infection. None of the 34 subjects who completed the treatment had virologic failure. Of the remaining seven subjects who did not achieve SVR12, five discontinued treatment on Day 1, one on Day 7, and one on Day 20 [see Adverse Reactions (6.1)].
16. How Supplied/Storage and Handling
Tablets
EPCLUSA tablets, 400 mg/100 mg, are pink, diamond-shaped, film-coated, debossed with "GSI" on one side and "7916" on the other. Each bottle contains 28 tablets (NDC 61958-2201-1), polyester coil, and is closed with a child-resistant closure.
EPCLUSA tablets, 200 mg/50 mg, are pink, oval-shaped, film-coated, debossed with "GSI" on one side and "S/V" on the other. Each bottle contains 28 tablets (NDC 61958-2203-1), polyester coil, and is closed with a child-resistant closure.
Oral Pellets
EPCLUSA oral pellets, 200 mg/50 mg, are white to off-white, film-coated pellets supplied as unit-dose packets in cartons. Each carton contains 28 packets (NDC 61958-2204-1).
EPCLUSA oral pellets, 150 mg/37.5 mg, are white to off-white, film-coated pellets supplied as unit-dose packets in cartons. Each carton contains 28 packets (NDC 61958-2205-1).
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Risk of Hepatitis B Virus Reactivation in Patients Coinfected with HCV and HBV
Inform patients that HBV reactivation can occur in patients coinfected with HBV during or after treatment of HCV infection. Advise patients to tell their healthcare provider if they have a history of HBV infection [see Warnings and Precautions (5.1)].
Serious Symptomatic Bradycardia When Coadministered with Amiodarone
Advise patients to seek medical evaluation immediately for symptoms of bradycardia such as near-fainting or fainting, dizziness or lightheadedness, malaise, weakness, excessive tiredness, shortness of breath, chest pain, confusion, or memory problems [see Warnings and Precautions (5.2), Adverse Reactions (6.2), and Drug Interactions (7.3)].
Drug Interactions
Inform patients that EPCLUSA may interact with other drugs. Advise patients to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products including St. John's wort [see Warnings and Precautions (5.2, 5.3) and Drug Interactions (7)].
Administration
Advise patients to take EPCLUSA once daily on a regular dosing schedule with or without food. Inform patients that it is important not to miss or skip doses and to take EPCLUSA for the duration that is recommended by the physician.
For EPCLUSA oral pellets, advise patients or caregivers to read and follow the Instructions for Use for preparing the correct dose [see Dosage and Administration (2.4, 2.5)].
Pregnancy
Advise patients to avoid pregnancy during combination treatment with EPCLUSA and ribavirin and for 6 months after completion of treatment. Inform patients to notify their healthcare provider immediately in the event of a pregnancy [see Use in Specific Populations (8.1)].
Spl Unclassified Section
Manufactured and distributed by:Gilead Sciences, Inc.Foster City, CA 94404
EPCLUSA, ATRIPLA, COMPLERA, GENVOYA, HARVONI, and STRIBILD are trademarks of Gilead Sciences, Inc., or its related companies. All other trademarks referenced herein are the property of their respective owners.
© 2022 Gilead Sciences, Inc. All rights reserved.208341-GS-010
Patient Package Insert
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 06/2021 | |
| PATIENT INFORMATION | ||
| EPCLUSA® (ep-KLOO-suh) (sofosbuvir and velpatasvir) tablets | EPCLUSA® (ep-KLOO-suh) (sofosbuvir and velpatasvir) oral pellets | |
| Important: If you take EPCLUSA with ribavirin, you should also read the Medication Guide for ribavirin. | ||
| What is the most important information I should know about EPCLUSA? EPCLUSA can cause serious side effects, including: Hepatitis B virus reactivation: Before starting treatment with EPCLUSA, your healthcare provider will do blood tests to check for hepatitis B virus infection. If you have ever had hepatitis B virus infection, the hepatitis B virus could become active again during or after treatment of hepatitis C virus with EPCLUSA. Hepatitis B virus becoming active again (called reactivation) may cause serious liver problems including liver failure and death. Your healthcare provider will monitor you if you are at risk for hepatitis B virus reactivation during treatment and after you stop taking EPCLUSA. For more information about side effects, see the section "What are the possible side effects of EPCLUSA?" | ||
| What is EPCLUSA? EPCLUSA is a prescription medicine used to treat adults and children 3 years of age and older with chronic (lasting a long time) hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5, or 6 infection:
It is not known if EPCLUSA is safe and effective in children under 3 years of age. | ||
Before taking EPCLUSA, tell your healthcare provider about all of your medical conditions, including if you:
Keep a list of your medicines to show your healthcare provider and pharmacist.
| ||
How should I take EPCLUSA?
| ||
How should I give EPCLUSA oral pellets to my child? See the detailed Instructions for Use for information about how to give or take a dose of EPCLUSA oral pellets.
| ||
| What are the possible side effects of EPCLUSA? EPCLUSA can cause serious side effects, including:
| ||
|
| |
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
How should I store EPCLUSA?
| ||
| General information about the safe and effective use of EPCLUSA Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use EPCLUSA for a condition for which it was not prescribed. Do not give EPCLUSA to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about EPCLUSA that is written for health professionals. | ||
| What are the ingredients in EPCLUSA? Active ingredients: sofosbuvir and velpatasvir Inactive ingredients, Tablets: copovidone, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablet film-coat contains: iron oxide red, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. Inactive ingredients, Oral Pellets: colloidal silicon dioxide, copovidone, croscarmellose sodium, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. The oral pellets film-coat contains: amino methacrylate copolymer, colloidal silicon dioxide, hypromellose, L‑tartaric acid, polyethylene glycol, stearic acid, talc, and titanium dioxide. Manufactured and distributed by: Gilead Sciences, Inc., Foster City, CA 94404 EPCLUSA is a trademark of Gilead Sciences, Inc., or its related companies. All other trademarks referenced herein are the property of their respective owners. ©2022 Gilead Sciences, Inc. All rights reserved. For more information, call 1-800-445-3235 or go to www.epclusa.com. 208341-GS-010 | ||
Instructions For Use Section
Read the Patient Information that comes with EPCLUSA oral pellets for important information about EPCLUSA.
This Instructions for Use contains information on how to take EPCLUSA oral pellets. Be sure you understand and follow the instructions. If you have any questions, ask your healthcare provider or pharmacist.
Important Information You Need to Know Before Taking EPCLUSA oral pellets
- For oral use only (take by mouth with or without food).
- Pellets should be swallowed whole. Do not chew pellets.
- Do not open the EPCLUSA oral pellet packet(s) until ready to use.
- For children younger than 6 years, take EPCLUSA oral pellets with food.
- EPCLUSA oral pellets are white to off-white pellets supplied as single-use packets in cartons. Each carton contains 28 packets.
- Do not use EPCLUSA oral pellets if the carton tamper-evident seal or the packet has been opened or damaged.
Gather Supplies:
Before you prepare a dose of EPCLUSA oral pellets, gather the following supplies:
- Daily EPCLUSA oral pellet packet(s), as prescribed by your healthcare provider
- Scissors (optional)
If taking EPCLUSA oral pellets with food, gather the following additional supplies:
- One or more spoonfuls of non-acidic soft food such as pudding, chocolate syrup, or ice cream
- Bowl
- Spoon
Prepare and Administer a Dose:
| Step 1: If taking EPCLUSA oral pellets with food, add one or more spoonfuls of non-acidic soft food to the bowl first. | ||
| Step 2: Hold the EPCLUSA oral pellets packet with the cut line on top (see Figure A). | Step 3: Shake the packet gently to settle the pellets to the bottom of the packet (see Figure B). | |
|
| |
| Step 4: Cut the packet along the cut line with scissors (see Figure C), or fold the packet back at the tear line (see Figure D) and tear open (see Figure E). | ||
 Figure C |  |  Figure D | 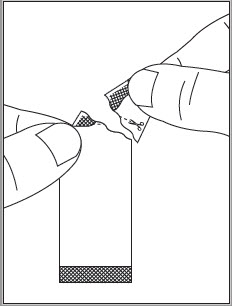 Figure E |
Step 5:
- If taking EPCLUSA oral pellets with food, carefully pour the entire contents of the prescribed number of EPCLUSA oral pellet packet(s) onto the food in the bowl and gently mix with a spoon (see Figure F). Take the EPCLUSA oral pellets and food mixture within 15 minutes without chewing to avoid a bitter taste.
- If taking EPCLUSA oral pellets by mouth, pour the entire contents of the prescribed number of EPCLUSA oral pellet packet(s) directly in the mouth and swallow without chewing to avoid a bitter taste (see Figure G). Water may be taken after swallowing the pellets, if needed.
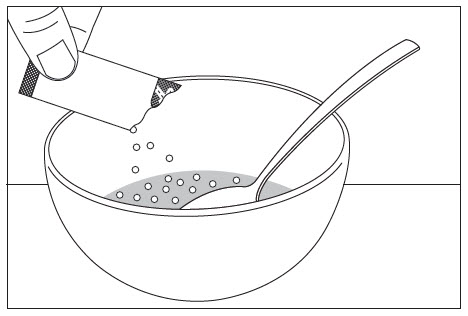 Figure F Figure F |  | 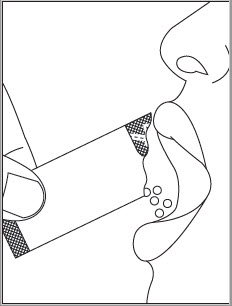 Figure G Figure G |
Step 6: Make sure all of the EPCLUSA oral pellets are taken. Make sure that no EPCLUSA oral pellets remain in the packet(s) or in the food.
Storing EPCLUSA oral pellets
- Store EPCLUSA pellets below 86°F (30°C).
- Keep EPCLUSA oral pellets and all medicines out of the reach of children.
Disposing of EPCLUSA oral pellets
- Throw away (dispose of) any unused portion. Do not store and reuse any leftover EPCLUSA oral pellets or EPCLUSA mixture (oral pellets mixed with food).
For more information, call 1-800-445-3235 or go to www.EPCLUSA.com.
Manufactured for and distributed by: Gilead Sciences, Inc., Foster City, CA 94404 EPCLUSA is a trademark of Gilead Sciences, Inc., or its related companies.
© 2022 Gilead Sciences, Inc. All rights reserved.208341-GS-010
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Issued: June 2021
Package Label. Principal Display Panel
Package Label. Principal Display Panel
Package Label. Principal Display Panel
Package Label. Principal Display Panel
NDC 61958-2205-128 packets
Rx only
Epclusa® (sofosbuvir and velpatasvir) oral pellets
150 mg/37.5 mg per packet
Pellets should be swallowed whole. Do not chew pellets.
Note to pharmacist: Do not cover ALERT box with pharmacy label.
ALERT: Find out about medicines that should NOT be taken with Epclusa
GILEAD



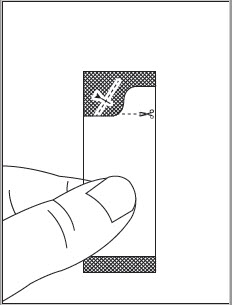 Figure A
Figure A Figure B
Figure B

