Health care personnel (HCP) are at risk for a variety of infectious pathogens following exposure to blood or body fluids.[1,2] Exposure to hepatitis C virus (HCV) is a well-recognized occupational risk for HCP, particularly following exposure to HCV-positive blood.[3] The following lesson outlines 2020 guidance from the Centers for Disease Control and Prevention (CDC) on the management of health care personnel potentially exposed to HCV.[3]
Definition of Health Care Personnel
The CDC defines HCP as all paid and unpaid persons providing health care or working or training in health care settings, who have reasonably anticipated risks for exposure to infectious materials, including blood or body fluids, contaminated medical supplies and equipment, or contaminated environmental surfaces.[4] Health care personnel may include, but are not limited to the following (in alphabetical order):
- Autopsy personnel
- Contractual staff not employed by the health care facility
- Dental personnel
- Emergency medical service personnel
- Laboratory personnel
- Nurses
- Nursing assistants
- Persons not directly involved in patient care but potentially exposed to blood and body fluids.(e.g., housekeeping, laundry, security, maintenance, and volunteers)
- Pharmacists
- Physicians
- Students and trainees
- Technicians
- Therapists
Sharps-Related Injuries
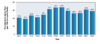
There are an estimated 385,000 sharps-related injuries each year among HCP working in hospitals in the United States.[5,6,7] Although this equates to more than 1,000 injuries per day, it is likely an underestimate, as it does not include sharps-related injuries sustained in other health care settings, such as nursing home facilities, clinics, and at an individual's home. Although data suggest the rate of hospital-based percutaneous exposures declined markedly following the 2001 Needlestick Safety and Prevention Act (and following changes in safety standards implemented by the Occupational Safety and Health Administration [OSHA]), 2022 data from 40 U.S. hospitals reported to the Exposure Prevention Information Network (EPINet) reveal that occupational needlestick and sharp object injuries exposures remain common, with 29.2 injuries per 100 average daily census days among reporting hospitals (Figure 1).[3,4,8] Percutaneous exposure is particularly common among trainees, with an estimated 18% of trainees sustaining a percutaneous exposure annually, most often from needles intended for intramuscular or subcutaneous injections or from suture needles.[4,9,10,11,12,13] Mucosal exposures are also common and occur in approximately 22% of trainees per year, but only 17% of those with a mucosal exposure reported the exposure to occupational health.[4]