Ascites is defined as an abnormal accumulation of fluid in the abdominal cavity. It is the most common complication of cirrhosis, with approximately 50% of persons with compensated cirrhosis developing ascites over the course of 10 years. After developing ascites that necessitates hospitalization, the risk of mortality increases to 15% at 1 year and nearly 50% at 5 years. Complications following the development of ascites include spontaneous bacterial peritonitis, dilutional hyponatremia, refractory ascites, hepatic hydrothorax, and hepatorenal syndrome. Development of these complications markedly decreases the likelihood of survival (Figure 1).[1,2] The development of ascites should prompt a referral for consideration of liver transplantation.
- Module 3 Overview
Management of Cirrhosis-Related Complications - 0%Lesson 1
Diagnosis and Management of AscitesActivities- 0%Lesson 2
Recognition and Management of Spontaneous Bacterial PeritonitisActivities- 0%Lesson 3
Screening for Varices and Prevention of BleedingLesson 1. Diagnosis and Management of Ascites
Learning Objective Performance Indicators
- Provide a list of causes in the differential diagnosis of ascites
- Diagnose ascites based on physical examination findings
- Describe the appropriate technique for performing abdominal paracentesis
- Summarize appropriate medical management and dietary recommendations for persons with ascites
- Discuss ascites-associated complications and treatment
Last Updated: March 4th, 2024Author:Jennifer Price, MD, PhDJennifer Price, MD, PhD
Associate Professor of Medicine
Director, UCSF Viral Hepatitis Center
Division of GI and Hepatology
University of California, San FranciscoDisclosures:- Grants, Research support to Institution: Abbvie, Genentech, Vir Biotechnology, Zydus Pharmaceuticals
Reviewers:Maria A. Corcorran, MD, MPH,Maria A. Corcorran, MD, MPH
Assistant Professor
Division of Allergy & Infectious Diseases
University of WashingtonDavid H. Spach, MDDavid H. Spach, MD
Professor of Medicine
Division of Allergy & Infectious Diseases
University of WashingtonDisclosures: NoneTable of ContentsBackground
Evaluation of Ascites
Causes of Ascites
In the United States, approximately 85% of persons with ascites have cirrhosis as the cause, with the other 15% resulting from a non-hepatic cause of fluid accumulation (Figure 2).[2] Approximately 5% of persons with cirrhosis have “mixed” ascites or have two or more causes for the ascites, typically cirrhosis plus another reason. In addition to assessing for risk factors for liver disease, history or risk factors for malignancy, heart failure, nephrotic syndrome, thyroid myxedema, recent abdominal surgery, and tuberculosis should be elicited.
Physical Examination
The presence of bulging flanks suggests the presence of ascites (Figure 3).[3] In order for the flank dullness to be appreciated on physical examination, at least 1,500 mL of ascites needs to be present. The shifting dullness test improves the diagnostic sensitivity of physical examination for detecting the presence of ascites; this test has 83% sensitivity and 56% specificity in detecting ascites.[3] Abdominal imaging should be performed when ascites is suspected on history and physical examination. A complete abdominal ultrasound will confirm the presence of ascites, may reveal evidence of cirrhosis and portal hypertension (e.g., nodular liver, portal vein diameter greater than 12 mm, or splenomegaly), and can assess for evidence of hepatocellular carcinoma or portal vein thrombosis.
Diagnostic and Therapeutic Paracentesis
An evaluation for the etiology of clinically apparent ascites should be done. In addition, a diagnostic paracentesis should be done during any hospitalization to assess for infection. Fasting is not required for this procedure. Prophylactic blood products, including fresh frozen plasma and platelets, do not routinely need to be given prior to paracentesis in persons with cirrhosis with associated thrombocytopenia and coagulopathy.[4] The tests for coagulation do not reflect the true bleeding risk in these individuals, as there is diminished production of both procoagulants and anticoagulants. There are no threshold criteria for coagulation parameters or platelet count for paracentesis. This procedure, however, should be avoided in the setting of clinically evident hyperfibrinolysis or disseminated intravascular coagulation. The following summarizes the key steps in performing an abdominal paracentesis.
- Body Position and Site for Paracentesis: The procedure is usually performed with the individual lying supine. The left lower quadrant of the abdomen is the preferred site for the paracentesis and the exact insertion site should be located 2 fingerbreadths (3 cm) cephalad and 2 fingerbreadths (3 cm) medial to the anterior superior iliac spine.[5] Some experts choose the midline of the abdomen midway between the pubis and umbilicus, but this site is considered less preferable in obese individuals (due to the increase in midline wall thickness) and in persons with lower volume ascites (a smaller pool of fluid in the midline than in the lateral quadrant). The right lower quadrant approach may be complicated by a dilated cecum or appendectomy scar. Extreme care should be taken to avoid the inferior epigastric arteries, which are located halfway between the pubis and anterior superior iliac spines and run cephalad in the rectus sheath, as well as visible collaterals in the abdominal wall. In addition, caution is needed in persons who have a palpable spleen, as it could be ruptured with the left lower quadrant approach. If the ascitic fluid is difficult to find on physical examination or if there is significant bowel dilatation, ultrasonography can be used to help locate the fluid pocket and visualize the spleen and other structures to guide this procedure. Paracentesis sites should be chosen distant from abdominal surgical scars or under image guidance.(Figure 4)
- Choosing Needle for Insertion: A 1.0- or 1.5-inch 21- or 22-gauge single-hole needle (or a 3.5-inch 22-gauge needle for obese persons) can be used for a diagnostic paracentesis, whereas a 15- or 16-gauge multi-hole two-piece needle set can be used for therapeutic paracentesis, involving the removal of more than 5 L of ascites for symptomatic relief from abdominal pain, early satiety, and/or dyspnea.
- Preparation and Insertion Technique: The site should be cleansed with iodine or chlorhexidine solution, and the skin should be anesthetized using 1% lidocaine solution via a 25- or 27-gauge needle. Sterile gloves should be worn to avoid contamination of samples. After raising a wheal in the superficial skin, 3 to 5 mL of lidocaine is used to anesthetize the soft tissue tract using the Z-track technique (the skin is pulled downward with the non-dominant hand while inserting the needle with the other hand (Figure 5). The purpose of doing the Z-track technique is to decrease the risk of ascitic fluid leak. The skin is not released until the needle enters the peritoneal cavity, indicated by the aspiration of ascitic fluid. The paracentesis needle is inserted along the same line using the Z-track technique. A scalpel can be used to create a skin nick to facilitate the entry of a larger gauge needle for therapeutic paracentesis. After entry into the peritoneum, the angle and depth of the paracentesis needle should be stabilized. The suction applied should be intermittent rather than continuous to avoid pulling in omentum or bowel into the needle tip and obstructing flow. If the flow of liquid stops, the person undergoing the procedure can be slowly repositioned in an effort to pool more fluid near the needle tip.
- Fluid Collection and Samples: For a diagnostic tap, a minimum of 25 mL of fluid should be collected. One to two mL of ascitic fluid should be injected into a purple top (EDTA) tube for the cell count and differential tests. Three to four mL of fluid should be directed into a red top tube for chemical analysis. Fluid should be directly inoculated into blood culture bottles at the bedside, typically 10 mL into each bottle. If needed, an additional 50 mL of fluid can be sent in a sterile syringe or cup for cytology or other tests. Vacuum bottles are used to assist the speed of fluid removal when performing a therapeutic paracentesis.
- Paracentesis Complications: The paracentesis procedure is generally very safe, with only a 1% risk of abdominal wall hematoma and a less than a 0.5% risk of mortality, even in persons with coagulopathy related to liver disease.[6] Post-paracentesis ascitic fluid leak can occur in 5% of individuals, especially when larger needles are used. More serious complications, such as hemoperitoneum and bowel perforation, are extremely rare, reported in less than 1 in 1000 cases.[7] Infections due to this procedure are rare, most often occurring in cases of bowel injury.[8]
Analysis of Ascitic Fluid
The following includes a summary of major laboratory tests to consider performing with diagnostic paracentesis. Other tests not discussed can be ordered if there is suspicion for alternative or additional causes of ascites. For any initial diagnostic paracentesis to evaluate ascites, it is important to determine whether portal hypertension is present and whether the ascitic fluid is infected.
- Albumin and Protein: For an initial diagnostic procedure, an ascitic fluid sample should be sent for albumin and total protein. The serum-ascites albumin gradient (SAAG) is calculated by subtracting the ascitic fluid albumin value from the serum albumin value obtained on the same day. A serum-ascites albumin gradient value greater than or equal to 1.1 g/dL is indicative of portal hypertension but does not exclude additional causes of ascites in a person with portal hypertension.[9] An ascitic fluid total protein value less than 2.5 g/dL is consistent with ascites from cirrhosis or nephrotic syndrome, whereas a high ascitic fluid protein value greater than 2.5 g/dL is seen in persons who have a cardiac cause of ascites.
- Cell Count and Cultures: Routinely, a cell count and differential should be performed on ascitic fluid. With any concern for infection, the fluid should be directly inoculated into aerobic and anaerobic blood culture bottles at the bedside prior to the administration of antibiotics, as it increases the yield of bacterial growth in culture from 50% to around 80% when the polymorphonuclear leukocyte (PMN) count is greater than or equal to 250 cells/mm3.[10,11] The yield on Gram’s staining of ascitic fluid is very low, except in the setting of bowel perforation into the ascites. Fungal cultures should be obtained if indicated.
- Glucose and Lactate Dehydrogenase: Ascitic glucose and lactate dehydrogenase (LDH) levels should be part of the analysis of ascitic fluid if secondary bacterial peritonitis is suspected. The ascitic fluid glucose level is typically similar to a serum glucose level, except in the setting of malignancy or infection. Findings that support a diagnosis of secondary bacterial peritonitis caused by gastrointestinal perforation include an ascitic glucose of less than 50 mg/dL, lactate dehydrogenase greater than 225 mU/mL, total protein greater than 1 g/dL, and polymicrobial infection.[12]
- Mycobacterial Smear and Culture: Ascitic fluid smear and culture for mycobacteria should be reserved for individuals at high risk for tuberculous peritonitis as the sensitivity of the smear is poor, and the sensitivity of the fluid culture for mycobacteria is only approximately 50%. The 4 to 6 weeks needed before culture results are available delays diagnosis. Ascitic fluid polymerase chain reaction (PCR) assays can be done, but the utility of these tests has not been well established. The gold standard for the diagnosis of tuberculous peritonitis remains directed peritoneal biopsy via laparoscopy or mini-laparotomy and mycobacterial culture.
- Cytology: Ascitic fluid cytology is expensive and is only revealing in the setting of peritoneal carcinomatosis, typically in persons with a history of breast, colon, gastric, or pancreatic carcinoma. At least 50 mL of fresh warm ascitic fluid needs to be immediately processed for optimal yield, with a sensitivity of 82.8% with one sample sent, improving to 96.7% when 3 samples are sent from different paracenteses.[13]
- Cancer Antigen 125: Serum cancer antigen 125 (CA125) can be elevated in any person with ascites or pleural effusion of any cause, as the level rises when mesothelial cells are under pressure in the presence of fluid, so it does not necessarily indicate ovarian malignancy in this setting. Thus, CA125 is not routinely ordered as a diagnostic test when evaluating ascitic fluid.
Persistent Ascites due to Cirrhosis
Individuals who undergo serial outpatient therapeutic paracenteses only need to have the ascitic fluid sample routinely sent for cell count and differential. At time of any hospital admission, before initiation of antibiotics, all persons with cirrhosis and ascites should undergo diagnostic paracentesis for cell count and differential and bacterial culture to assess for spontaneous bacterial peritonitis. The diagnosis of spontaneous bacterial peritonitis requires an elevated ascitic fluid absolute PMN count of greater than or equal to 250 cells/mm3 without an obvious treatable intraabdominal source of infection, which should prompt empiric antibiotic therapy.[14,15]
Basic Management of Ascites
The following summarizes key recommendations in the 2021 AASLD Guidance for Management of Ascites.[2] In general, sodium restriction and diuretics are the mainstays of treatment for persons with ascites due to portal hypertension, but individuals with low SAAG (less than 1.1 g/dL) ascites do not respond well to these measures.[2]
Treatment of the Underlying Disorder
Cessation of alcohol use is vital to the management of ascites due to alcohol-associated liver disease. In one study of hospitalized persons with Child-Turcotte-Pugh class C cirrhosis due to severe alcohol-associated liver disease, 75% of those who remained abstinent were still alive at 3 years whereas most who continued to drink alcohol were not.[16] Treatment of autoimmune hepatitis or chronic viral hepatitis can also lead to significant clinical improvement and resolution of ascites in some cases. Similar to the management of liver-related ascites, treatment of ascites in non-hepatic cases should focus on treatment of the underlying disorder (e.g., treatment of tuberculosis, treatment of secondary bacterial peritonitis, or surgical resection of benign ovarian tumor).
Dietary Sodium Restriction
Individuals with portal hypertension-associated ascites should restrict their daily dietary sodium intake to less than 2,000 mg (90 mmol).[2] Further restriction risks malnutrition due to poor palatability of foods. A 24-hour urinary sodium excretion can be measured to assess the adequacy of fluid loss and dietary sodium restriction. Completeness of the 24-hour collection is estimated by measurement of 24-hour urinary creatinine; accounting for some anticipated loss of body mass in the setting of cirrhosis, daily excretion of creatinine should exceed 15 mg/kg body weight in cirrhotic men and 10 mg/kg body weight in cirrhotic women. The goal of treatment is to increase the daily urinary excretion of sodium to a value above 80 mmol per day, so that in conjunction with daily nonurinary sodium excretion, the daily sodium excretion should exceed the allowed daily dietary intake of sodium.[2] Random urinary sodium concentration is not useful because of the variable sodium excretion throughout the day, but a random “spot” urine sodium/potassium ratio correlates with 24-hour urinary sodium excretion, with higher ratios indicating greater urinary excretion. Thus, a ratio of greater than one is desired. Persons who are excreting a sufficient amount of urinary sodium (24-hour urinary sodium greater than 80 mmol per day or spot urine sodium/potassium ratio greater than one) and are not losing weight are likely consuming more than 2,000 mg of sodium daily and need further education and adherence counseling. On the other hand, the diuretic dose should be increased in persons not excreting a sufficient amount of urinary sodium, unless they are diuretic refractory.
Fluid Restriction
Dietary sodium restriction is more important than fluid restriction in the management of cirrhosis. Fluid restriction is not necessary unless the serum sodium concentration is less than 125 mmol/L or mental status changes attributed to hyponatremia develop.[2] Rapid correction of chronic hyponatremia (with hypertonic saline or other means) should be avoided due to risk of osmotic demyelination syndrome.
Diuretics
In persons with portal hypertension, the combination of spironolactone and furosemide, starting at doses of 100 mg daily and 40 mg daily, respectively, is recommended.[2] In older persons or persons weighing 50 kg or less, lower starting doses of 50 mg daily and 20 mg daily, respectively, are typically used. Single-agent spironolactone can be used and is superior to single-agent furosemide,[17] but combination therapy leads to more rapid fluid loss in persons with moderate ascites and decreases the risk of hyperkalemia. If weight loss is insufficient while maintaining the 100:40 mg ratio, the doses of the diuretics may be increased simultaneously every 3 to 5 days to maximum daily doses of 400 mg of spironolactone and 160 mg of furosemide. The combined single morning dosing improves compliance, optimizes diuresis, and avoids nocturia. A ratio less than 100:40 mg of spironolactone and furosemide may be used for persons with parenchymal renal disease with concern for hyperkalemia. Furosemide can be temporarily held or reduced for those with hypokalemia.
Option if Intolerant to Spironolactone
For individuals unable to tolerate spironolactone due to painful gynecomastia, amiloride (10 to 40 mg daily) can be substituted, although it has a lower natriuretic effect than spironolactone.[18] Eplerenone is an aldosterone antagonist used to treat heart failure and is not associated with gynecomastia but has not been extensively studied yet for the management of ascites.[19] Hydrochlorothiazide in combination with furosemide is not recommended due to combined hypokalemia. Torsemide and bumetanide have also been used in combination with spironolactone in the management of ascites, but they have not demonstrated superiority over furosemide.
Daily Limit for Weight Loss
In persons with significant peripheral edema, there is no limit for daily weight loss, but in those without peripheral edema, daily weight loss should be restricted to 0.5 kg maximum. Diuretics may need to be held in the setting of significant volume loss such as active gastrointestinal hemorrhage or diarrhea, uncontrolled or recurrent hepatic encephalopathy, significant hyponatremia (serum sodium less than 120 mmol/L) despite fluid restriction, or renal dysfunction (e.g., serum creatinine greater than 2.0 mg/dL).
Medications to Avoid
The use of angiotensin-converting-enzyme inhibitors and angiotensin-receptor blockers should be avoided in persons with cirrhosis, due to concerns of renal failure and increased mortality for those who develop hypotension. Hypotension independently predicts increased one-year mortality in persons with cirrhosis. Among individuals with refractory ascites, propranolol is associated with decreased survival, perhaps due to the increased risk of paracentesis-induced circulatory dysfunction, so the risks and benefits of its use should be considered individually for each person.[20] Nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin, should also be avoided due to the risk of reduced urinary sodium excretion and renal failure. Although vaptans can improve hyponatremia, there are significant risks associated with use of these types of agents in persons with cirrhosis. For example, tolvaptan, a selective oral vasopressin V2-receptor antagonist used to treat hypervolemic and euvolemic hyponatremia, has been shown to be effective in persons with refractory ascites, but is contraindicated for use in persons with underlying liver disease, including those with cirrhosis, due to risk of causing severe hepatotoxicity.[21,22] Moreover, hyponatremia recurs upon discontinuation of the medication.[23] Satavaptan was evaluated for the management of ascites in persons with cirrhosis and was potentially associated with a higher risk of mortality.[24]
Management of Tense Ascites
A single large-volume paracentesis followed by dietary sodium restriction and initiation of diuretics is appropriate as initial therapy for new-onset large-volume ascites.[25] Up to 5 liters can be removed without significant disturbances in systemic and renal hemodynamics,[26] but if more than 5 liters of ascitic fluid is removed, then intravenous albumin (8 g/L of fluid removed) should be given.[27]
Management of Refractory Ascites
Among persons with cirrhosis and ascites, fewer than 10% will develop refractory ascites, which is defined as ascites that is unresponsive to dietary sodium restriction and maximal diuretic dosing (typically, spironolactone 400 mg daily and furosemide 160 mg daily), or that recurs rapidly after therapeutic paracentesis.[28] There are two different subtypes: diuretic-resistant ascites (lack of response to dietary sodium restriction and intensive diuretic treatment) and diuretic-intractable ascites (diuretic-induced complications such as hepatic encephalopathy, renal insufficiency, hyponatremia, or hyperkalemia that prevent optimization of diuretic dosing). Once refractory ascites develops, one-year mortality is approximately 50%. Options for treatment include optimization of medical management, serial large volume paracenteses, transjugular intrahepatic portosystemic shunt (TIPS), peritoneovenous shunt, and liver transplantation.
Medication Considerations
As mentioned previously, propranolol has been shown to be associated with decreased survival in the setting of refractory ascites and discontinuation should be considered. Angiotensin-converting-enzyme inhibitors and angiotensin-receptor blockers should be avoided. Oral midodrine, an agent used to treat hypotension, was shown to increase mean arterial pressure and improve survival in a pilot study that enrolled persons with cirrhosis who had refractory or recurrent ascites.[29] There are insufficient data to routinely recommend midodrine use in this setting.
Serial Large-Volume Therapeutic Paracenteses
Once an individual is deemed diuretic refractory, diuretics should be discontinued, and management may rely upon serial large-volume therapeutic paracenteses alone. Typically, a large-volume paracentesis (up to 8 to 10 L removed) performed every 2 weeks should control ascites in a person who is adherent with dietary sodium restriction.[30] Need for more frequent paracenteses suggests dietary noncompliance. The use of indwelling intraabdominal catheters is reserved for persons with malignancy-associated ascites and is not recommended in this situation. Long-term serial paracenteses can lead to significant loss of protein and worsen malnutrition, but placement of a percutaneous endoscopic gastrostomy (PEG) tube in an effort to provide nutrition should be avoided in these individuals due to the high risk of mortality associated with performing the procedure.[31]
Albumin Infusions with Therapeutic Paracentesis
In one randomized study, the use of intravenous albumin (10 grams administered per liter of fluid removed) in the setting of therapeutic paracentesis decreased the risk of negative changes in plasma renin and serum creatinine levels.[32] A meta-analysis of 17 trials demonstrated a reduction in risk of post-paracentesis circulatory dysfunction, hyponatremia, and mortality in the albumin group (odds ratio of death 0.64, 95% CI, 0.41-0.98); study protocols typically used a 20% or higher concentration of albumin solution, and administered 5 to 10 g of albumin per liter of fluid removed.[27] With a large-volume paracentesis (5 liters or more removed), some experts recommend giving 6 to 8 g of intravenous albumin for every liter of ascitic fluid removed, with the albumin infused during or immediately following the paracentesis. In the United States, both 5% and 25% concentrations of intravenous albumin are available, but the 25% solution is preferred since the 5% solution would deliver 5 times the amount of sodium for the equivalent amount of albumin.
Transjugular Intrahepatic Portosystemic Shunt (TIPS)
A transjugular intrahepatic portosystemic shunt (TIPS), which is placed by Interventional Radiology, has been shown in multiple multicenter, randomized, controlled trials to be superior to serial large-volume paracenteses in the control of ascites, but with varying results on the impact on overall transplant-free survival and the risk of inducing or worsening hepatic encephalopathy.[33,34,35,36,37,38] Polytetrafluoroethylene-covered stents are preferred over uncovered stents due to decreased rates of TIPS occlusion.
- Absolute Contraindications: The absolute contraindications to placement of TIPS include congestive heart failure (particularly right-sided heart failure), severe tricuspid regurgitation, severe pulmonary hypertension (mean pulmonary pressure greater than 45 mmHg), extensive polycystic liver disease, and uncontrolled infection or biliary obstruction (Figure 6).[33]
- Relative Contraindications: The relative contraindications for performing TIPS include obstruction of all hepatic veins, complete portal vein thrombosis, hepatocellular carcinoma (especially if centrally located), severe coagulopathy (international normalized ratio [INR] greater than 5) or thrombocytopenia (platelet count less than 20,000/cm3), moderate pulmonary hypertension, recurrent or persistent severe spontaneous hepatic encephalopathy, advanced liver failure (bilirubin greater than 5 mg/dL or Model for End-stage Liver Disease [MELD] score greater than 17), cardiac dysfunction (ejection fraction less than 60%), cardiac diastolic dysfunction, and advanced age (e.g., greater than 69 years) (Figure 7).[33]
- Outcome after TIPS: Short- and long-term mortality rates following TIPS can be estimated using MELD and Child-Turcotte-Pugh scoring systems. Clinical improvement in ascites following TIPS occurs in 74% of patients.[39] Diuretics may need to be continued even after the placement of TIPS. Approximately 30% of persons develop hepatic encephalopathy after TIPS, though most can be managed medically (e.g., lactulose). Risk factors for the development of hepatic encephalopathy after TIPS include older age and a history of pre-TIPS hepatic encephalopathy.[40] A small-diameter stent of less than 10 mm is preferred to reduce the likelihood of hepatic encephalopathy after TIPS.[2] Narrowing or occluding the TIPS can treat severe debilitating hepatic encephalopathy resistant to medical therapy, which, fortunately, is rare. Those with renal dysfunction, especially those on dialysis, may have a reduced response to TIPS.
Peritoneovenous Shunts
The use of peritoneovenous shunts for management of ascites has fallen out of favor due to limited long-term patency (less than 20% at 2 years), risk of complications, and no improvement in survival compared to medical therapy.[41,42,43] It is reserved as palliative treatment in select individuals who are not candidates for transplantation, TIPS, or serial therapeutic paracenteses.[2]
Complications Associated with Ascites
Spontaneous Bacterial Peritonitis (SBP)
The diagnosis of SBP requires an ascitic fluid absolute polymorphonuclear count greater than or equal to 250 cells/mm3 without an obvious intraabdominal, surgically-treatable source and should prompt empiric antibiotic treatment with an intravenous third-generation cephalosporin, preferably cefotaxime 2 g every 8 hours, for 5 days.[44,45] Due to increasing multidrug resistance, diagnostic paracentesis should be repeated after 48 hours to assess treatment response. In addition, broader initial antibiotic therapy is recommended in persons at higher risk of multidrug-resistant organisms.[2] A recommended broader-spectrum regimen consists of piperacillin-tazobactam, with vancomycin added in persons who have prior SBP or a positive surveillance culture for methicillin-resistant Staphylococcus aureus (MRSA).[2] For persons with prior SBP or a positive surveillance swab for vancomycin-resistant enterococcus, daptomycin should be used instead of vancomycin.[2] Also, meropenem, with or without vancomycin, should be used instead of piperacillin-tazobactam in persons with current or recent piperacillin-tazobactam use.[2] Individuals with SBP who have a serum creatinine greater than 1 mg/dL, blood urea nitrogen greater than 30 mg/dL, or total bilirubin greater than 4 mg/dL should receive intravenous albumin 1.5 g per kg body weight upon diagnosis and 1.0 g per kg body weight on day 3 after diagnosis.[2,46,47] After an episode of SBP, long-term prophylaxis with daily norfloxacin or trimethoprim-sulfamethoxazole is indicated.[48] More detailed information regarding diagnosis, treatment, and prevention of SBP is provided in the lesson Recognition and Management of Spontaneous Bacterial Peritonitis in this same Module.
Hyponatremia
The most common type of hyponatremia is hypervolemic (dilutional) hyponatremia. This occurs because vasodilatation in cirrhosis triggers activation of the renin-angiotensin system and sympathetic nervous system, leading to avid sodium and water retention with increased antidiuretic hormone release, resulting in dilutional hyponatremia. Some patients with cirrhosis can also develop hypovolemic hyponatremia, most often secondary to diuretics and laxatives. Up to 50% of patients with cirrhosis and ascites have a serum sodium concentration of less than 135 mmol/L. Hyponatremia is an independent risk factor for morbidity and mortality in persons with cirrhosis, and serum sodium has been added to the original MELD scoring system for liver transplant prioritization.[1,18,49]
- Indication for the Treatment of Hyponatremia: Treatment specifically for hyponatremia is not necessary unless the serum sodium concentration drops below 120 mmol/L, which occurs in only 1% of persons, or if there are neurologic symptoms attributed to hyponatremia. If treated, the rate of correction should not exceed an increase of more than 9 mmol/L per day, with a goal of increasing only 4 to 6 mmol/L per day, in order to avoid the risk of osmotic demyelination syndrome.[50]
- Approach to Treatment of Hyponatremia: In general, over-rapid correction of hyponatremia before, during, and after liver transplantation should be avoided. The management of hyponatremia should take into account whether the hyponatremia is hypovolemic, euvolemic, or hypervolemic.[2] Euvolemic hyponatremia is uncommon and should be managed by addressing the underlying cause.
- Treatment of Hypovolemic Hyponatremia: The key aspects to treating hypovolemic hyponatremia in persons with cirrhosis are to discontinue diuretics and/or laxatives, in conjunction with providing fluid resuscitation. The fluid resuscitation should consist of 5% intravenous albumin or a crystalloid (ideally Lactated Ringer’s solution).[2]
- Treatment of Hypervolemic Hyponatremia: The management of hypervolemic hyponatremia should include a multipronged approach that should take into account the following items.[2]
- Fluid Restriction: Relative fluid restriction (1,000 mL per day) should be a component of first-line treatment in persons who have ascites and a serum sodium less than 126 mEq/L; true fluid restriction (total fluid intake less than urine volume) is difficult to achieve. If the hyponatremia is severe (less than 120 mEq/L), even more severe fluid restriction may be needed. Less severe fluid restriction is usually needed for adequately managing persons with mild hyponatremia (serum sodium 126-135 mEq/L).
- Discontinuation of Diuretics and Laxatives: Diuretics and laxatives should be reduced in dose or discontinued if possible. In this situation, if hypotension is present, beta-blockers may also need to be reduced in dose or discontinued.
- Albumin Infusion: The administration of an albumin infusion, 1 g/kg body weight with a maximum of 100 g/day, is generally recommended along with fluid restriction if the hyponatremia is severe (serum sodium less than 120 mEq/L).
- Management of Concomitant Hypokalemia: Treatment of hypokalemia may also assist in raising serum sodium concentration.
- Use of Vasopressin Receptor Antagonists: Vasopressin receptor antagonists (vaptans) cause selective water diuresis and raise serum sodium concentrations but are not routinely used in persons with cirrhosis. Conivaptan is a V1a receptor blocker that requires intravenous administration and is not recommended in persons with cirrhosis because of the concern that it can increase the risk of hypotension and renal compromise. Tolvaptan, an oral V2 receptor blocker, should be used with extreme caution in persons with cirrhosis due to concerns for liver injury. This side effect was observed in a clinical study of tolvaptan in persons with polycystic kidney disease but was not noted among patients with cirrhosis. The vaptan agents should be used with caution and not for more than 30 days, due to the possible risk of hepatocellular injury.
- Hypertonic Saline: In general, hypertonic saline should be avoided in persons with cirrhosis, except for short-term correction, such as to partially correct severe hyponatremia to a serum sodium concentration above 125 mEq/L immediately prior to liver transplantation, or in those with symptomatic hyponatremia.
Acute Kidney Injury and Hepatorenal Syndrome
Approximately 20-50% of hospitalized individuals with cirrhosis and ascites will develop some type of renal dysfunction. Acute kidney injury (AKI) is associated with a poor prognosis and is an independent predictor of mortality in patients with cirrhosis. Hepatorenal syndrome (HRS) is a type of AKI, now termed HRS-AKI.[51]
- Diagnosis and Stages of AKI: AKI is defined by the International Club of Ascites‐Acute Kidney Injury criteria as an increase in serum creatinine greater than or equal to 0.3 mg/dL within 48 hours OR at least a 50% increase in serum creatinine above baseline over 7 days. AKI is classified into three stages.[2]
Table 1. Stages of Acute Kidney Injury
Acute Kidney Inury Stage Description Stage 1* Increase of creatinine ≥0.3 mg/dL up to 2-fold of baseline Stage 2 Increase in creatinine between 2-fold and 3-fold of baseline Stage 3 Increase in creatinine >3-fold of baseline or creatinine >4 mg/dL (353.6 μmol/L) with an acute increase ≥0.3 mg/dL (26.5 μmol/L) or initiation of renal replacement therapy *Within stage 1, the absolute level of serum creatinine has clinical significance. For example, patients with AKI stage 1 with serum creatinine ≥1.5 mg at diagnosis fared significantly worse than those with lower serum creatinine. Some members of the writing group favored adopting literature proposing stage 1A (creatinine <1.5 mg/dL) and stage 1B (creatinine ≥1.5 mg/dL). The rest of the group felt the effect of creatinine on the patient outcome is continuous.
Source:- Biggins SW, Angeli P, Garcia-Tsao G, et al. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1014-48. [PubMed Abstract]
- Diagnostic Criteria for HRS-AKI: The diagnostic criteria for hepatorenal syndrome requires all the following: (1) cirrhosis with ascites, (2) AKI as defined by the International Club of Ascites‐Acute Kidney Injury criteria, (3) no response after at least 2 consecutive days with diuretic withdrawal and volume expansion with albumin (recommended dose is 1 g/kg body weight per day), (4) absence of shock, (5) no current or recent treatment with nephrotoxic drugs, and (6) absence of parenchymal kidney disease, as indicated by proteinuria greater than 500 mg per day, microhematuria (greater than 50 red blood cells per high-power field), and/or abnormal renal ultrasonography.[2,52]
Table 2. Diagnosis of Hepatorenal Syndrome-Acute Kidney Injury (HRS-AKI)*
Cirrhosis with ascites - Diagnosis of acute kidney injury (AKI) according to International Club of Ascites-Acute Kidney Injury† criteria
- No response after 2 consecutive days of diuretic withdrawal and plasma volume expansion with albumin infusion (1 g/kg body weight per day)
- Absence of shock
- No current or recent use of nephrotoxic drugs (nonsteroidal anti-inflammatory drugs, aminoglycosides, or iodinated contrast media)
- No signs of structural kidney injury, as indicated by proteinuria (>500 mg per day), microhematuria (>50 red blood cells per high-power field), and/or abnormal renal ultrasonography
*The old terminology, type-1 HRS, has been replaced by HRS-AKI. For reference, therapeutic studies to date have used the historical definition: sudden impairment of kidney function, namely a 100% increase in serum creatinine to a value >2.5 mg/dL (221 μmol/L) within <2 weeks.
†Increase in serum creatinine ≥0.3 mg/dL from baseline within 48 hours or a percent increase in serum creatinine of ≥50% which is known or presumed to have occurred within the preceding 7 days.Source:- Biggins SW, Angeli P, Garcia-Tsao G, et al. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1014-48. [PubMed Abstract]
- Classification of Hepatorenal Syndrome: Prior to the new AKI criteria, there were two types of hepatorenal syndrome: type 1 and type 2.[53] Type 1 hepatorenal syndrome was characterized by rapidly progressive renal failure with a doubling in the initial serum creatinine to a level greater than 2.5 mg/dL (or 50% reduction in the initial 24-hour creatinine clearance to a level lower than 20 mL/min) in less than 2 weeks. Type 1 hepatorenal syndrome now falls under the definition of HRS-AKI. It is frequently triggered by a precipitating event, such as SBP, alcoholic hepatitis, urinary tract infection, or intravascular volume contraction, and is associated with acute rapid deterioration of circulatory function with hypotension and activation of endogenous vasoconstrictor systems, and leads to a very poor prognosis, with a median survival of around 2 weeks in untreated individuals. Type 2 hepatorenal syndrome was typically associated with refractory ascites and is characterized by a slower, progressive decline in renal function, typically with a serum creatinine that ranges from 1.5 to 2.5 mg/dL, and a median survival of 4 to 6 months. Type 2 hepatorenal syndrome now falls under the definition of chronic kidney disease (CKD), which is defined as an estimated glomerular filtration rate less than 60 mL/1.73m2 per minute for at least 3 months.
- Management of HRS-AKI: Management is focused on the treatment of the precipitating event, renal failure, and systemic inflammatory response syndrome. Measures to prevent HRS-AKI include the use of intravenous albumin in persons with SBP at high risk of developing hepatorenal syndrome and the use of prophylactic antibiotics in persons with cirrhosis and gastrointestinal bleeding.[54] Once HRS-AKI syndrome is established, diuretics should be discontinued and vasoconstrictors used to decrease systemic vasodilatation and improve renal perfusion. The combination of terlipressin and albumin has been shown to be superior to albumin alone and placebo for the treatment of HRS-AKI and may be effective in more than 30% of cases.[55,56] Where terlipressin is not available, midodrine, an alpha-agonist, is used instead (titrated up to 15 mg three times daily) in combination with octreotide, starting with 100 mcg subcutaneously three times daily, titrated up to 200 mcg three times daily and albumin (1 g/kg bolus, up to 100 g) on day 1 followed by 25 to 50 g daily until midodrine and octreotide therapy are discontinued), with a goal of increasing mean arterial pressure by 10 to 15 mm Hg to a level above 82 mm Hg. This combination achieves a response rate of around 30% and responders typically continue treatment for at least 2 weeks.[57] For persons in the intensive care unit, continuous norepinephrine infusion (0.5 to 3.0 mg/h) combined with intravenous albumin boluses for at least 2 days can be considered as well for HRS-AKI, with a goal of raising mean arterial blood pressure by 10 mm Hg.[58,59] In addition, in select individuals who fail to respond to medical therapy, TIPS can be used to improve renal function, but should be avoided in persons with advanced liver dysfunction. Ultimately, liver transplantation is the definitive treatment for this condition, and some even require renal replacement therapy as a bridge to transplantation.[60]
Umbilical Hernia
Up to 20% of individuals with cirrhosis and ascites can develop umbilical hernias. Complications related to these hernias include omental or bowel strangulation, typically after paracentesis or shunt procedure, and hernia perforation.[61] Individuals with ascites and an umbilical hernia should wear an abdominal binder to minimize strain and enlargement of the hernia and should be educated on the warning symptoms of an incarcerated hernia. Preemptive TIPS should be considered to prevent rupture of thin-walled umbilical hernias.[62,63] The risks and benefits of elective surgical repair need to be assessed individually. Among persons who are medical candidates for surgery (e.g., Child-Turcotte-Pugh class A cirrhosis), the ascites needs to be controlled first with optimal medical management or TIPS; otherwise, the hernia will recur in over 70% of individuals.[64] Emergent surgical repair due to incarceration or rupture should be performed by surgeons experienced with persons who have cirrhosis. If feasible, TIPS should be considered before or after the surgery, along with dietary sodium and fluid restriction.
Hepatic Hydrothorax
Approximately 5 to 10% of individuals with cirrhosis and ascites develop hepatic hydrothorax, which is typically a right-sided pleural effusion.[65] It is a result of fluid being drawn up from the peritoneal cavity into the pleural space through small defects in the diaphragm. Sometimes, minimal to almost no fluid remains in the abdomen. Injection of technetium-radiolabeled sulfur colloid into the abdomen followed by transdiaphragmatic flow of the isotope into the thoracic space can confirm ascites as the origin of the pleural effusion, if needed.[66] Thoracentesis does not require platelet or fresh frozen plasma transfusions, and there is no data-supported limit for the amount of fluid that can be removed.[67] Due to differences in hydrostatic pressure, the protein concentration is higher in pleural fluid than ascites. Spontaneous bacterial empyema can occur in the absence of spontaneous bacterial peritonitis and can be treated with appropriate antibiotic therapy without placement of a chest tube.[68] Chest tube placement in persons with hepatic hydrothorax is associated with massive fluid losses, high morbidity (greater than 90%) and high mortality (greater than 30% in the absence of TIPS), so it should be avoided.[69,70] Treatment should start with dietary sodium restriction and diuretics. Therapeutic thoracentesis can be done for dyspnea. TIPS can be performed as treatment for refractory hepatic hydrothorax. Most individuals with hepatic hydrothorax are not good candidates for pleurodesis due to the rapid rate of fluid reaccumulation. Individuals with refractory hepatic hydrothorax who are not candidates for TIPS should be referred for consideration of liver transplantation.
Summary Points
- The development of ascites indicates decompensation of cirrhosis and should prompt an immediate referral for liver transplantation.
- Prophylactic blood products do not need to be administered prior to paracentesis, even in the setting of coagulopathy or thrombocytopenia, but paracentesis should be avoided in persons with disseminated intravascular coagulation or untreated hyperfibrinolysis.
- A SAAG of greater than or equal to 1.1 g/dL indicates portal hypertension as the cause of ascites, with cirrhosis or heart failure being common causes of portal hypertension. Additional diagnostic tests can be ordered based on clinical suspicion.
- Treatment of ascites in persons with cirrhosis should be focused on dietary sodium restriction of less than 2,000 mg daily and the use of diuretics, specifically, spironolactone and furosemide, titrated using a respective ratio of 100:40 mg. Fluid restriction is reserved only for those with a serum sodium concentration of less than 125 mmol/L or symptomatic hyponatremia.
- Treatment options for the management of refractory ascites include optimization of medical therapy, serial large-volume therapeutic paracenteses with the use of intravenous albumin, TIPS in select candidates, and liver transplantation. Peritoneovenous shunt is typically reserved only for individuals who are not candidates for the other therapies.
- An ascitic fluid absolute polymorphonuclear count greater than or equal to 250 cells/mm3 should prompt empiric antibiotic treatment for spontaneous bacterial peritonitis with intravenous cefotaxime (2 g every 8 hours) for five days.
- Individuals with untreated Type 1 hepatorenal syndrome have very poor short-term survival and should be referred for urgent liver transplant evaluation.
- In most circumstances, placement of a chest tube is contraindicated in persons with hepatic hydrothorax due to risk of massive fluid loss and high morbidity and mortality.
Citations
- 1.Planas R, Montoliu S, Ballesté B, et al. Natural history of patients hospitalized for management of cirrhotic ascites. Clin Gastroenterol Hepatol. 2006;4:1385-94.[PubMed Abstract] -
- 2.Biggins SW, Angeli P, Garcia-Tsao G, et al. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1014-48.[PubMed Abstract] -
- 3.Cattau EL Jr, Benjamin SB, Knuff TE, Castell DO. The accuracy of the physical examination in the diagnosis of suspected ascites. JAMA. 1982;247:1164-6.[PubMed Abstract] -
- 4.Northup PG, Garcia-Pagan JC, Garcia-Tsao G, et al. Vascular Liver Disorders, Portal Vein Thrombosis, and Procedural Bleeding in Patients With Liver Disease: 2020 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;73:366-413.[PubMed Abstract] -
- 5.Sakai H, Sheer TA, Mendler MH, Runyon BA. Choosing the location for non-image guided abdominal paracentesis. Liver Int. 2005;25:984-6.[PubMed Abstract] -
- 6.De Gottardi A, Thévenot T, Spahr L, Morard I, Bresson-Hadni S, Torres F, Giostra E, Hadengue A. Risk of complications after abdominal paracentesis in cirrhotic patients: a prospective study. Clin Gastroenterol Hepatol. 2009;7:206-9.[PubMed Abstract] -
- 7.Pache I, Bilodeau M. Severe haemorrhage following abdominal paracentesis or ascites in patients with liver failure. Aliment Pharmacol Ther. 2005;21:525-9.[PubMed Abstract] -
- 8.Runyon BA. Paracentesis of ascitic fluid: a safe procedure. Arch Intern Med. 1986;146:2259-61.[PubMed Abstract] -
- 9.Runyon BA, Montano AA, Akriviadis EA, Antillon MR, Irving MA, McHutchison JG. The serum-ascites albumin gradient is superior to the exudate-transudate concept in the differential diagnosis of ascites. Ann Intern Med. 1992;117:215-20.[PubMed Abstract] -
- 10.Runyon BA, Antillon MR, Akriviadis EA, McHutchinson JG. Bedside inoculation of blood culture bottles is superior to delayed inoculation in the detection of spontaneous bacterial peritonitis. J Clin Microbiol. 1990;28:2811-2.[PubMed Abstract] -
- 11.Runyon BA, Canawati HN, Akriviadis EA. Optimization of ascitic fluid culture technique. Gastroenterology. 1988;95:1351-5.[PubMed Abstract] -
- 12.Runyon BA, Hoefs JC. Ascitic fluid analysis in the differentiation of spontaneous bacterial peritonitis from gastrointestinal tract perforation into ascitic fluid. Hepatology. 1984;4:447-50.[PubMed Abstract] -
- 13.Runyon BA. Malignancy-related ascites and ascitic fluid “humoral tests of malignancy.” J Clin Gastroenterol. 1994;18:94-8.[PubMed Abstract] -
- 14.Runyon BA. Management of adult patients with ascites due to cirrhosis: an update. Hepatology. 2009;49:2087-107.[PubMed Abstract] -
- 15.Such J, Runyon BA. Spontaneous bacterial peritonitis. Clin Infect Dis. 1998;27:669-74.[PubMed Abstract] -
- 16.Veldt BJ, Lainé F, Guillygomarc’h A, et al. Indication of liver transplantation in severe alcoholic liver cirrhosis: quantitative evaluation and optimal timing. J Hepatol. 2002;36:93-8.[PubMed Abstract] -
- 17.Pérez-Ayuso RM, Arroyo V, Planas R, et al. Randomized comparative study of efficacy of furosemide versus spironolactone in nonazotemic cirrhosis with ascites. Relationship between the diuretic response and the activity of the renin-aldosterone system. Gastroenterology. 1983;84:961-8.[PubMed Abstract] -
- 18.Angeli P, Dalla Pria M, De Bei E, Albino G, Caregaro L, Merkel C, Ceolotto G, Gatta A. Randomized clinical study of the efficacy of amiloride and potassium canrenoate in nonazotemic cirrhotic patients with ascites. Hepatology. 1994;19:72-9.[PubMed Abstract] -
- 19.Dimitriadis G, Papadopoulos V, Mimidis K. Eplerenone reverses spironolactone-induced painful gynaecomastia in cirrhotics. Hepatol Int. 2011;5:738-9.[PubMed Abstract] -
- 20.Sersté T, Melot C, Francoz C, et al. Deleterious effects of beta-blockers on survival in patients with cirrhosis and refractory ascites. Hepatology. 2010;52:1017-22.[PubMed Abstract] -
- 21.Zhang X, Wang SZ, Zheng JF, et al. Clinical efficacy of tolvaptan for treatment of refractory ascites in liver cirrhosis patients. World J Gastroenterol. 2014;20:11400-5.[PubMed Abstract] -
- 22.Ohki T, Sato K, Yamada T, et al. Efficacy of tolvaptan in patients with refractory ascites in a clinical setting. World J Hepatol. 2015;7:1685-93.[PubMed Abstract] -
- 23.Cárdenas A, Ginès P, Marotta P, et al. Tolvaptan, an oral vasopressin antagonist, in the treatment of hyponatremia in cirrhosis. J Hepatol. 2012;56:571-8.[PubMed Abstract] -
- 24.Wong F, Watson H, Gerbes A, et al. Satavaptan for the management of ascites in cirrhosis: efficacy and safety across the spectrum of ascites severity. Gut. 2012;61:108-16.[PubMed Abstract] -
- 25.Ginès P, Arroyo V, Quintero E, et al. Comparison of paracentesis and diuretics in the treatment of cirrhotics with tense ascites. Results of a randomized study. Gastroenterology. 1987;93:234-41.[PubMed Abstract] -
- 26.Peltekian KM, Wong F, Liu PP, Logan AG, Sherman M, Blendis LM. Cardiovascular, renal and neurohumoral responses to single large-volume paracentesis patients with cirrhosis and diuretic-resistant ascites. Am J Gastroenterol. 1997;92:394-9.[PubMed Abstract] -
- 27.Bernardi M, Caraceni P, Navickis RJ, Wilkes MM. Albumin infusion in patients undergoing large-volume paracentesis: a meta-analysis of randomized trials. Hepatology. 2012;55:1172-81.[PubMed Abstract] -
- 28.Arroyo V, Ginès P, Gerbes AL, et al. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. International Ascites Club. Hepatology. 1996;23:164-76.[PubMed Abstract] -
- 29.Singh V, Dhungana SP, Singh B, et al. Midodrine in patients with cirrhosis and refractory or recurrent ascites: a randomized pilot study. J Hepatol. 2012;56:348-54.[PubMed Abstract] -
- 30.Titó L, Ginès P, Arroyo V, et al. Total paracentesis associated with intravenous albumin management of patients with cirrhosis and ascites. Gastroenterology. 1990;98:146-51.[PubMed Abstract] -
- 31.Baltz JG, Argo CK, Al-Osaimi AM, Northup PG. Mortality after percutaneous endoscopic gastrostomy in patients with cirrhosis: a case series. Gastrointest Endscop. 2010;72:1072-5.[PubMed Abstract] -
- 32.Ginès P, Titó L, Arroyo V, et al. Randomized comparative study of therapeutic paracentesis with and without intravenous albumin in cirrhosis. Gastroenterology. 1988;94;1493-502.[PubMed Abstract] -
- 33.Boyer TD, Haskal ZJ, American Association for the Study of Liver Diseases. The Role of Transjugular Intrahepatic Portosystemic Shunt (TIPS) in the Management of Portal Hypertension: update 2009. Hepatology. 2010;51:306.[PubMed Abstract] -
- 34.Ginès P, Uriz J, Calahorra B, et al. Transjugular intrahepatic portosystemic shunting versus paracentesis plus albumin for refractory ascites in cirrhosis. Gastroenterology. 2002;123:1839-47.[PubMed Abstract] -
- 35.Rössle M, Ochs A, Gülberg V, et al. A comparison of paracentesis and transjugular intrahepatic portosystemic shunting in patients with ascites. N Engl J Med. 2000;342:1701-7.[PubMed Abstract] -
- 36.Salerno F, Cammà C, Enea M, Rössle M, Wong F. Transjugular intrahepatic portosystemic shunt for refractory ascites: a meta-analysis of individual patient data. Gastroenterology. 2007;133:825-34.[PubMed Abstract] -
- 37.Salerno F, Merli M, Riggio O, et al. Randomized controlled study of TIPS versus paracentesis plus albumin in cirrhosis with severe ascites. Hepatology. 2004;40:629-35.[PubMed Abstract] -
- 38.Sanyal AJ, Genning C, Reddy KR, et al. The North American Study for the Treatment of Refractory Ascites. Gastroenterology. 2003;124:634-41.[PubMed Abstract] -
- 39.Somberg KA, Lake JR, Tomlanovich SJ, LaBerge JM, Feldstein V, Bass NM. Transjugular intrahepatic portosystemic shunts for refractory ascites: assessment of clinical and hormonal response and renal function. Hepatology. 1995;21:709-16.[PubMed Abstract] -
- 40.Sanyal AJ, Freedman AM, Shiffman ML, Purdum PP 3rd, Luketic VA, Cheatham AK. Portosystemic encephalopathy after transjugular intrahepatic portosystemic shunt: results of a prospective controlled study. Hepatology. 1994;20:46-55.[PubMed Abstract] -
- 41.Ginès P, Arroyo V, Vargas V, et al. Paracentesis with intravenous infusion of albumin as compared with peritoneovenous shunting in cirrhosis with refractory ascites. N Engl J Med. 1991;325:829-35.[PubMed Abstract] -
- 42.Guardiola J, Xiol X, Escribá JM, et al. Prognosis assessment of cirrhotic patients with refractory ascites treated with a peritoneovenous shunt. Am J Gastroenterol. 1995;90:2097-102.[PubMed Abstract] -
- 43.Stanley MM, Ochi S, Lee KK, et al. Peritoneovenous shunting as compared with medical treatment in patients with alcoholic cirrhosis and massive ascites. Veterans Administration Cooperative Study on Treatment of Alcoholic Cirrhosis with Ascites. N Engl J Med. 1989;321:1632-8.[PubMed Abstract] -
- 44.Hoefs JC, Canawati HN, Sapico FL, Hopkins RR, Weiner J, Montgomerie JZ. Spontaneous bacterial peritonitis. Hepatology. 1982;2:399-407.[PubMed Abstract] -
- 45.Runyon BA, McHutchinson JG, Antillon MR, Akriviadis EA, Montano A. Short-course versus long-course antibiotic treatment of spontaneous bacterial peritonitis: a randomized controlled trial of 100 patients. Gastroenterology. 1991;100:1737-42.[PubMed Abstract] -
- 46.Sigal SH, Stanca CM, Fernandez J, Arroyo V, Navasa M. Restricted use of albumin for spontaneous bacterial peritonitis. Gut. 2007;56:597-9.[Gut] -
- 47.Sort P, Navasa M, Arroyo V, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med. 1999;341:403-9.[PubMed Abstract] -
- 48.Ginès P, Rimola A, Plana R, et al. Norfloxacin prevents spontaneous bacterial peritonitis recurrence in cirrhosis: results of a double-blind, placebo-controlled trial. Hepatology. 1990;12:716-24.[PubMed Abstract] -
- 49.Heuman DM, Abou-Assi SG, Habib A, et al. Persistent ascites and low serum sodium to identify patients with cirrhosis and low MELD scores who are high risk for early death. Hepatology. 2004;40:802-10.[PubMed Abstract] -
- 50.Gankam Kengne F, Decaux G. Hyponatremia and the Brain. Kidney Int Rep. 2018;3:24-35.[PubMed Abstract] -
- 51.Ginès P, Solà E, Angeli P, Wong F, Nadim MK, Kamath PS. Author Correction: Hepatorenal syndrome. Nat Rev Dis Primers. 2018;4:33.[PubMed Abstract] -
- 52.Salerno F, Gerbes A, Ginès P, Wong F, Arroyo V. Diagnosis, prevention and treatment of hepatorenal syndrome in cirrhosis. Gut. 2007;56:1310-8.[PubMed Abstract] -
- 53.Egerod Israelsen M, Gluud LL, Krag A. Acute kidney injury and hepatorenal syndrome in cirrhosis. J Gastroenterol Hepatol. 2015;30:236-43.[PubMed Abstract] -
- 54.Ginès P, Guevera M, Arroyo V, Rodés J. Hepatorenal syndrome. Lancet. 2003;362:1819-27.[PubMed Abstract] -
- 55.Martín-Llahí M, Pépin MN, Guevara M, et al. Terlipressin and albumin vs albumin in patients with cirrhosis and hepatorenal syndrome: a randomized study. Gastroenterology. 2008;134:1352-9.[PubMed Abstract] -
- 56.Sanyal AJ, Boyer T, Garcia-Tsao G, et al. A randomized, prospective, double-blind, placebo-controlled trial of terlipressin for type 1 hepatorenal syndrome. Gastroenterology. 2008;134:1360-8.[PubMed Abstract] -
- 57.Esrailian E, Pantangco ER, Kyulo NL, Hu KQ, Runyon BA. Octreotide/Midodrine therapy significantly improves renal function and 30-day survival in patients with type 1 hepatorenal syndrome. Dig Dis Sci. 2007;52:742-8.[PubMed Abstract] -
- 58.Gluud LL, Christensen K, Christensen E, Krag A. Systematic review of randomized trials on vasoconstrictor drugs for hepatorenal syndrome. Hepatology. 2010;51:576-84.[PubMed Abstract] -
- 59.Duvoux C, Zanditenas D, Hézode C, et al. Effects of noradrenalin and albumin in patients with type I hepatorenal syndrome: a pilot study. Hepatology. 2002;36:374-80.[PubMed Abstract] -
- 60.Wong F. Recent advances in our understanding of hepatorenal syndrome. Nat Rev Gastroenterol Hepatol. 2012;9:382-91.[PubMed Abstract] -
- 61.Belghiti J, Durand F. Abdominal wall hernias in the setting of cirrhosis. Semin Liver Dis. 1997;17:219-26.[PubMed Abstract] -
- 62.Telem DA, Schiano T, Divino CM. Complicated hernia presentation in patients with advanced cirrhosis and refractory ascites: management and outcome. Surgery. 2010;148:538-43.[PubMed Abstract] -
- 63.Triantos CK, Kehagias I, Nikolopoulou V, Burrhoughs AK. Surgical repair of umbilical hernias in cirrhosis with ascites. Am J Med Sci. 2011;341:222-6.[PubMed Abstract] -
- 64.Runyon BA, Juler GL. Natural history of repaired umbilical hernias in patients with and without cirrhosis. Am J Gastroenterol. 1985;80:38-9.[PubMed Abstract] -
- 65.Strauss RM, Boyer TD. Hepatic hydrothorax. Semin Liver Dis. 1997;17:227-32.[PubMed Abstract] -
- 66.Rubinstein D, McInnes IE, Dudley FJ. Hepatic hydrothorax in the absence of clinical ascites: diagnosis and management. Gastroenterology. 1985;88:188-91.[PubMed Abstract] -
- 67.Xiol X, Castellote J, Cortes-Beut R, Delgado M, Guardiola J, Sesé E. Usefulness and complications of thoracentesis in cirrhotic patients. Am J Med. 2001;111:67-9.[Elsevier] -
- 68.Xiol X, Castellví JM, Guardiola J, Sesé E, Castellote J, Perelló A, Cervantes X, Iborra MJ. Spontaneous bacterial empyema in cirrhotic patients: a prospective study. Hepatology. 1996;23: 719-23.[PubMed Abstract] -
- 69.Orman ES, Lok AS. Outcomes of patients with chest tube insertion for hepatic hydrothorax. Hepatol Int. 2009;3:582-6.[PubMed Abstract] -
- 70.Runyon BA, Greenblatt M, Ming RH. Hepatic hydrothorax is a relative contraindication to chest tube insertion. Am J Gastroenterol. 1986;81:566-7.[PubMed Abstract] -
Additional References
- Angeli P, Fasolato S, Mazza E, et al. Combined versus sequential diuretic treatment of ascites in non-azotaemic patients with cirrhosis: results of an open randomised clinical trial. Gut. 2010;59:98-104.[PubMed Abstract] -
- Angeli P, Wong F, Watson H, Ginès P; CAPPS Investigators. Hyponatremia in cirrhosis: Results of a patient population survey. Hepatology. 2006;44:1535-42.[PubMed Abstract] -
- Bajaj JS, Tandon P, OʼLeary JG, et al. The Impact of Albumin Use on Resolution of Hyponatremia in Hospitalized Patients With Cirrhosis. Am J Gastroenterol. 2018;113:1339.[PubMed Abstract] -
- Gunawan B, Runyon B. The efficacy and safety of epsilon-aminocaproic acid treatment in patients with cirrhosis and hyperfibrinolysis. Aliment Pharmacol Ther. 2006;23:115-20.[PubMed Abstract] -
- Llach J, Ginès P, Arroyo V, et al. Prognostic value of arterial pressure, endogenous vasoactive systems, and renal function in cirrhotic patients admitted to the hospital for the treatment of ascites. Gastroenterology. 1988;94:482-7.[PubMed Abstract] -
- Miller PD, Linas SL, Schrier RW. Plasma demeclocycline levels and nephrotoxicity. Correlation in hyponatremic cirrhotic patients. JAMA. 1980;243;2513-5.[PubMed Abstract] -
- Montoliu S, Ballesté B, Planas R, et al. Incidence and prognosis of different types of functional renal failure in cirrhotic patients with ascites. Clin Gastroenterol Hepatol. 2010;8:616-22.[PubMed Abstract] -
- Pockros PJ, Reynolds TB. Rapid diuresis in patients with ascites from chronic liver disease: the importance of peripheral edema. Gastroenterology. 1986;90:1827-33.[PubMed Abstract] -
- Runyon BA, Hoefs JC, Canawati HN. Polymicrobial bacterascites. A unique entity in the spectrum of infected ascitic fluid. Arch Intern Med. 1986;146:2173-5.[PubMed Abstract] -
- Santos J, Planas R, Pardo A, et al. Spironolactone alone or in combination with furosemide in the treatment of moderate ascites in nonazotemic cirrhosis. A randomized comparative study of efficacy and safety. J Hepatol. 2003;39:187-92.[PubMed Abstract] -
- Schrier RW, Gross P, Gheorghiade M, et al. Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N Engl J Med. 2006;355:2099-112.[PubMed Abstract] -
- Singh V, Dheerendra PC, Singh B, et al. Midodrine versus albumin in the prevention of paracentesis-induced circulatory dysfunction in cirrhotics: a randomized pilot study. Am J Gastroenterol. 2008;103:1399-405.[PubMed Abstract] -
- Wong F, Blei AT, Blendis LM, Thuluvath PJ. A vasopressin receptor antagonist (VPA-985) improves serum sodium concentration in patients with hyponatremia: a multicenter, randomized, placebo-controlled trial. Hepatology. 2003;37:182-91.[PubMed Abstract] -
- Wu SS, Lin OS, Chen Y-Y, Hwang KL, Soon MS, Keeffe EB. Ascitic fluid carcinoembryonic antigen and alkaline phosphatase levels for the differentiation of primary from secondary bacterial peritonitis with intestinal perforation. J Hepatol. 2001;34:215-21.[PubMed Abstract] -
Figures
 Figure 1. Natural History and Survival of Persons with AscitesThis figure shows the 1- and 5-year survival of persons with ascites. Patients who do not develop complications have markedly better survival than those who develop dilutional hyponatremia, refractory ascites, or hepatorenal syndrome.Source: Planas R, Montoliu S, Ballesté B, Rivera M, Miquel M, Masnou H, Galeras JA, Giménez MD, Santos J, Cirera I, Morillas RM, Coll S, Solà R. Natural history of patients hospitalized for management of cirrhotic ascites. Clin Gastroenterol Hepatol. 2006;4:1385-94.
Figure 1. Natural History and Survival of Persons with AscitesThis figure shows the 1- and 5-year survival of persons with ascites. Patients who do not develop complications have markedly better survival than those who develop dilutional hyponatremia, refractory ascites, or hepatorenal syndrome.Source: Planas R, Montoliu S, Ballesté B, Rivera M, Miquel M, Masnou H, Galeras JA, Giménez MD, Santos J, Cirera I, Morillas RM, Coll S, Solà R. Natural history of patients hospitalized for management of cirrhotic ascites. Clin Gastroenterol Hepatol. 2006;4:1385-94. Figure 2. Differential Diagnosis of AscitesAbbreviations: SAAG = serum-ascites albumin gradient; SBP = spontaneous bacterial peritonitis; CHF = congestive heart failure; LDH = lactate dehydrogenase; CEA = carcinoembryonic antigen.Source: Runyon BA, Montano AA, Akriviadis EA, Antillon MR, Irving MA, McHutchinson JG. The serum-ascites albumin gradient is superior to the exudate-transudate concept in the differential diagnosis of ascites. Ann Intern Med. 1992; 117:215-20.
Figure 2. Differential Diagnosis of AscitesAbbreviations: SAAG = serum-ascites albumin gradient; SBP = spontaneous bacterial peritonitis; CHF = congestive heart failure; LDH = lactate dehydrogenase; CEA = carcinoembryonic antigen.Source: Runyon BA, Montano AA, Akriviadis EA, Antillon MR, Irving MA, McHutchinson JG. The serum-ascites albumin gradient is superior to the exudate-transudate concept in the differential diagnosis of ascites. Ann Intern Med. 1992; 117:215-20. Figure 3 (Image Series). Ascites: Physical Examination FindingsThis illustration shows an individual and ascites manifested on physical examination by bulging flanks.
Figure 3 (Image Series). Ascites: Physical Examination FindingsThis illustration shows an individual and ascites manifested on physical examination by bulging flanks.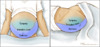 Figure 3B. Shifting Dullness in Person with AscitesTo perform the shifting dullness test, have the individual move to a supine position, then percuss the entire abdominal region, and mark the dullness-tympany transition point (left figure). Then place the person in the right lateral decubitus position, wait 30 to 60 seconds, repeat the percussion, and again mark the dullness-tympany transition point (right figure). A positive shifting dullness test is indicated by a shifting of the transition point.
Figure 3B. Shifting Dullness in Person with AscitesTo perform the shifting dullness test, have the individual move to a supine position, then percuss the entire abdominal region, and mark the dullness-tympany transition point (left figure). Then place the person in the right lateral decubitus position, wait 30 to 60 seconds, repeat the percussion, and again mark the dullness-tympany transition point (right figure). A positive shifting dullness test is indicated by a shifting of the transition point.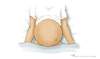 Figure 4 (Image Series). Paracentesis SiteIn most situations, the preferred site for performing a diagnostic paracentesis is the left lower quadrant. The midline region is not considered as safe due to the epigastric arteries in this region.
Figure 4 (Image Series). Paracentesis SiteIn most situations, the preferred site for performing a diagnostic paracentesis is the left lower quadrant. The midline region is not considered as safe due to the epigastric arteries in this region.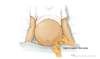 Figure 4B. Identifying Paracentesis SiteTo identify the preferred region for paracentesis in the left lower quadrant, first locate the anterior superior iliac spine. Then, mark a spot 2 fingerbreadths (3 cm) cephalad and 2 fingerbreadths (3 cm) medial to the anterior superior iliac spine.
Figure 4B. Identifying Paracentesis SiteTo identify the preferred region for paracentesis in the left lower quadrant, first locate the anterior superior iliac spine. Then, mark a spot 2 fingerbreadths (3 cm) cephalad and 2 fingerbreadths (3 cm) medial to the anterior superior iliac spine. Figure 4C. Inferior Epigastric ArteriesThe region of the inferior epigastric arteries should be avoided during paracentesis due to risk of arterial rupture if punctured during the procedure.
Figure 4C. Inferior Epigastric ArteriesThe region of the inferior epigastric arteries should be avoided during paracentesis due to risk of arterial rupture if punctured during the procedure.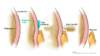 Figure 5. Paracentesis Z-Track TechniqueThe paracentesis Z technique is performed to minimize the risk of a peritoneal fluid leak. The Z-track technique consists of pulling the skin down by approximately 2 centimeters before inserting and advancing the needle. After the needle has been inserted, the skin is released. The concept is that the punctured hole in the skin, muscle, and fascia do not entirely overlap if the z technique is used.
Figure 5. Paracentesis Z-Track TechniqueThe paracentesis Z technique is performed to minimize the risk of a peritoneal fluid leak. The Z-track technique consists of pulling the skin down by approximately 2 centimeters before inserting and advancing the needle. After the needle has been inserted, the skin is released. The concept is that the punctured hole in the skin, muscle, and fascia do not entirely overlap if the z technique is used. Figure 6. Absolute Contraindications to Performing Transjugular Intrahepatic Portosystemic Shunt (TIPS) ProSource: Boyer TD, Haskal ZJ; American Association for the Study of Liver Diseases. The Role of Transjugular Intrahepatic Portosystemic Shunt (TIPS) in the Management of Portal Hypertension: update 2009. Hepatology. 2010;51:306.
Figure 6. Absolute Contraindications to Performing Transjugular Intrahepatic Portosystemic Shunt (TIPS) ProSource: Boyer TD, Haskal ZJ; American Association for the Study of Liver Diseases. The Role of Transjugular Intrahepatic Portosystemic Shunt (TIPS) in the Management of Portal Hypertension: update 2009. Hepatology. 2010;51:306. Figure 7. Relative Contraindications to Performing Transjugular Intrahepatic Portosystemic Shunt (TIPS) ProcedureSource: Boyer TD, Haskal ZJ; American Association for the Study of Liver Diseases. The Role of Transjugular Intrahepatic Portosystemic Shunt (TIPS) in the Management of Portal Hypertension: update 2009. Hepatology. 2010;51:306.
Figure 7. Relative Contraindications to Performing Transjugular Intrahepatic Portosystemic Shunt (TIPS) ProcedureSource: Boyer TD, Haskal ZJ; American Association for the Study of Liver Diseases. The Role of Transjugular Intrahepatic Portosystemic Shunt (TIPS) in the Management of Portal Hypertension: update 2009. Hepatology. 2010;51:306.Tables
Table 1. Stages of Acute Kidney Injury
Acute Kidney Inury Stage Description Stage 1* Increase of creatinine ≥0.3 mg/dL up to 2-fold of baseline Stage 2 Increase in creatinine between 2-fold and 3-fold of baseline Stage 3 Increase in creatinine >3-fold of baseline or creatinine >4 mg/dL (353.6 μmol/L) with an acute increase ≥0.3 mg/dL (26.5 μmol/L) or initiation of renal replacement therapy *Within stage 1, the absolute level of serum creatinine has clinical significance. For example, patients with AKI stage 1 with serum creatinine ≥1.5 mg at diagnosis fared significantly worse than those with lower serum creatinine. Some members of the writing group favored adopting literature proposing stage 1A (creatinine <1.5 mg/dL) and stage 1B (creatinine ≥1.5 mg/dL). The rest of the group felt the effect of creatinine on the patient outcome is continuous.
Source:- Biggins SW, Angeli P, Garcia-Tsao G, et al. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1014-48. [PubMed Abstract]
Table 2. Diagnosis of Hepatorenal Syndrome-Acute Kidney Injury (HRS-AKI)*
Cirrhosis with ascites - Diagnosis of acute kidney injury (AKI) according to International Club of Ascites-Acute Kidney Injury† criteria
- No response after 2 consecutive days of diuretic withdrawal and plasma volume expansion with albumin infusion (1 g/kg body weight per day)
- Absence of shock
- No current or recent use of nephrotoxic drugs (nonsteroidal anti-inflammatory drugs, aminoglycosides, or iodinated contrast media)
- No signs of structural kidney injury, as indicated by proteinuria (>500 mg per day), microhematuria (>50 red blood cells per high-power field), and/or abnormal renal ultrasonography
*The old terminology, type-1 HRS, has been replaced by HRS-AKI. For reference, therapeutic studies to date have used the historical definition: sudden impairment of kidney function, namely a 100% increase in serum creatinine to a value >2.5 mg/dL (221 μmol/L) within <2 weeks.
†Increase in serum creatinine ≥0.3 mg/dL from baseline within 48 hours or a percent increase in serum creatinine of ≥50% which is known or presumed to have occurred within the preceding 7 days.Source:- Biggins SW, Angeli P, Garcia-Tsao G, et al. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;74:1014-48. [PubMed Abstract]
Share by e-mail
Check
-On-
Learning
QuestionsThe Check-on-Learning Questions are short and topic related. They are meant to help you stay on track throughout each lesson and check your understanding of key concepts.You must be signed in to customize your interaction with these questions.
- 0%Lesson 2
Since you've received 80% or better on this quiz, you may claim continuing education credit.
You seem to have a popup blocker enabled. If you want to skip this dialog please Always allow popup windows for the online course.
 Elbasvir-Grazoprevir Zepatier
Elbasvir-Grazoprevir Zepatier Glecaprevir-Pibrentasvir Mavyret
Glecaprevir-Pibrentasvir Mavyret Ledipasvir-Sofosbuvir Harvoni
Ledipasvir-Sofosbuvir Harvoni Ribavirin Copegus, Rebetol, Ribasphere
Ribavirin Copegus, Rebetol, Ribasphere Sofosbuvir Sovaldi
Sofosbuvir Sovaldi Sofosbuvir-Velpatasvir Epclusa
Sofosbuvir-Velpatasvir Epclusa Sofosbuvir-Velpatasvir-Voxilaprevir Vosevi
Sofosbuvir-Velpatasvir-Voxilaprevir Vosevi








